Abstract
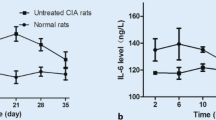
The collagen-induced arthritis model in rats was used to study the effect of disodium clodronate on inflammation and destruction of tarsal, metatarsal, and interphalangeal bones and joints. Female DA rats were immunized with heterologous type II collagen. Fourteen days after immunization, rats with similar scores were assigned to the different experimental groups. They were treated subcutaneously either with saline (controls) or with clodronate at doses of 12.5 and 25 mg/kg/day five times a week for 2 weeks. Clinical signs of arthritis including the severity of paw swelling were assessed weekly. At the time of killing, histological features of the non-decalcified tarsus with tarsal, tarsometatarsal and interphalangeal joints were assessed for inflammatory soft-tissue, articular, and bone changes. All the arthritic control rats developed severe arthritis as shown by the total histological scores of the hindpaw. The treatment with clodronate (25 mg/kg) decreased clinical signs of arthritis, the activity of the collagen-degrading lysosomal enzyme,β-N-acetylglucosaminidase, in inflamed hindpaw tissue, serum osteocalcin level and serum cross-linked telopeptide of type I collagen level. Histological evaluation indicated moderate arthritis in 29% of the rats and severe arthritis in 71%. The results show that clodronate given therapeutically to arthritic rats, induced with type II collagen, suppresses the intensity of inflammation and bone lesions in the tibiotarsal and tarsometatarsal regions.
Similar content being viewed by others
References
Trentham DE, Townes AS, Kang AH. Autoimmunity to type II collagen: an experimental model of arthritis. J Exp Med 1977;146:857–68.
Caulfield JP, Hein A, Dynesius-Trentham R, Trentham DE. Morphologic demonstration of two stages in the development of type II collagen-induced arthritis. Lab Invest 1982;46:321–43.
Trentham DE. Collagen arthritis as a relevant model for rheumatoid arthritis: evidence pro and con. Arthritis Rheum 1982;25:911–6.
Stuart JM, Townes AS, Kang AH. Collagen autoimmune arthritis. Ann Rev Immunol 1984;2:199–218.
Cremer MA, Townes AS, Kang AH. Collagen-induced arthritis in rodents. A review of clinical, histological and immunological features. Ryumachi 1984;24:45–56.
Terato K, Hashida R, Miyamoto K, Morimoto T, Kato Y, Kobayashi S, Tajima T, Otake S, Hori H, Nagai Y. Histological, immunological and biochemical studies on type II collagen-induced arthritis in rats. Biomed Res 1982;3:495–505.
Cannon GW, McCall S, Cole BC, Griffiths MM, Radov LA, Ward JR. Effects of indomethacin, cyclosporin, cyclophosphamide, and placebo on collagen-induced arthritis of mice. Agents Actions 1990;29:315–23.
Cannon GW, McCall S, Cole BC, Radov LA, Ward JR, Griffiths MM. Effects of gold sodium thiomalate, cyclosporin A, cyclosphosphamide, and placebo and collagen-induced arthritis in rats. Agents Actions 1993;38:240–6.
Flora L. Comparative antiinflammatory and bone protective effects of two diphosphonates in adjuvant arthritis. Arthritis Rheum 1979;22:340–6.
Barbier A, Breliere JC, Remandet B, Roncucci R. Studies on the chronic phase of adjuvant arthritis: effects of SR 41319, a new diphosphonate. Ann Rheum Dis 1986;45:67–74.
Francis MD, Hovancik K, Boyce RW. NE-58095: A diphosphonate which prevents bone erosion and preserves joint architecture in experimental arthritis. Int J Tissue React 1989;XI:239–52.
Österman T, Kippo K, Laurén L, Hannuniemi R, Sellman R. Effect of clodronate on established adjuvant arthritis. Rheumatol Int 1994;14:139–47.
Markusse HM, Lafeber GJM, Breedveld FC. Bisphosphonates in collagen arthritis. Rheumatol Int 1990;9:281–3.
Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983;16:109–10.
Vaes G, Jacques P. Studies on bone enzymes. Biochem J 1965;97:380–8.
Horak E, Hopfer SM, Sunderman FW. Spectrophotometric assay for urinary N-acetyl-β-D-glucosaminidase activity. Clin Chem 1981;27;1180–5.
Schenk RK, Olah AJ, Herrmann W. Preparation of calcified tissues for light microscopy. In: Dickson GR, editor. Methods of calcified tissue preparation. Elsevier: Amsterdam 1984;1–56.
Baron R, Vignery A, Neff L, Silverglate A, Santa Maria A. Processing of undecalcified bone specimens for bone histomorphometry. In: Becker RR, editor. Bone histomorphometry: techniques and interpretation. CRC Press, Boca Raton 1983;13–35.
Owen RT. Adjuvant induced polyarthritis — an overview. Methods Find Exp Clin Pharmacol 1980;2:199–204.
Taurog JD, Argentieri DC, McReunolds RA. Adjuvant Arthritis. In: Sabato GD, editor. Methods in enzymology, immunochemical techniques. Academic, San Diego, 1988; 339–55.
Hayward MA, Howard GA, Neuman RG, Wood DD, Weichman BM, Van Sickle DC. Prostaglandins in inflammatory bone pathology: mechanism and therapeutic benefit of etodolac. Agents Actions 1989;26:310–8.
Del Pozo E. Bone loss in adjuvant arthritis. Triangle 1989;28:43–50.
Weichman BM. Rat adjuvant arthritis: a model of chronic inflammation. In: Chang JY, Lewis AJ, editors. Pharmacological methods in the control of inflammation. Alan R Liss, New York 1989; 363–80.
Billingham MEJ. Models of arthritis and the search for antiarthritic drugs. Pharmacol Ther 1983;21:389–428.
Greenwald RA. Animal models for evaluation of arthritis drugs. Methods Find Exp Clin Pharmacol 1991;13:75–83.
Griffiths MM, DeWitt CW. Genetic control of collageninduced arthritis in rats: the immune response to type II collagen among susceptible and resistant strains and evidence for multiple gene control. J Immunol 1984;132:2830–6.
Holmdahl R, Vingsbo C, Hedrich H, Karlsson M, Kvick C, Goldschmith T, Gustafsson K. Homologous collagen-induced arthritis in rats and mice are associated with structurally different major histocompatibility complex DQ-like molecules. Eur J Immunol 1992;22:419–24.
Bataille R, Delmas PD, Chappard D, Sany J. Abnormal serum bone Gla protein levels in multiple myeloma: crucial role of bone formation and prognostic implications. Cancer 1990;66:167–72.
Hauschka PV, Lian JB, Cole DEC, Gundberg CM. Osteocalcin and matrix Gla protein: vitamin K-dependent proteins in bone. Physiol Rev 1989;69:990–1047.
Eriksen EF, Charles P, Melsen F, Mosekilde L, Risteli L, Risteli J. Serum markers of type I collagen formation and degradation in metabolic bone disease. Correlation to bone histomorphometry. J Bone Miner Res 1993;8:127–32.
Hakala M, Risteli L, Manelius J, Nieminen P, Risteli J. Increased type I collagen degradation correlates with disease severity in rheumatoid arthritis. Ann Rheum Dis 1993;52:866–9.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Österman, T., Kippo, K., Laurén, L. et al. Effect of clodronate on established collagen-induced arthritis in rats. Inflamm Res 44, 258–263 (1995). https://doi.org/10.1007/BF01782979
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01782979