Abstract
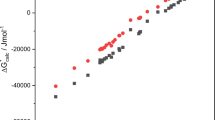
The SSOZ (site-site Ornstein-Zernike) equation with an original closure condition for liquid molecular systems is used to calculate thermodynamic functions of noble gas solvation in water. Water is modeled by two close sets of atom-atomic potential functions. The calculations indicate that the chemical solvation potential is strongly sensitive to water structure. A comparison with experiment is given.
Similar content being viewed by others
References
D. Chandler and H. C. Andersen,J. Chem. Phys.,57, No. 5, 1930–1946 (1972).
B. Pettitt and P. J. Rossky,,77, No. 3, 1451–1457 (1982).
P. A. Monson and G. P. Morris,Advances in Chemical Physics, I. Prigogin and S. Rice (eds.), Vol. LXXVII, 451–550 (1990).
H. Tanaka,J. Chem. Phys.,88, No. 3, 1512–1520 (1987).
H.-A. Yu, B. Roux, and M. Karplus,,92, No. 2, 5020–5030 (1990).
J. Perkyns and B. M. Pettitt,Biophys. Chem.,51, 129–146 (1994).
L. Lue and D. Blankschteln,J. Phys. Chem.,96, No. 21, 8582–8594 (1992).
G. A. Martynov and G. N. Sarkisov,Mol. Phys.,49, No. 6, 1495–1504 (1983).
D. A. Tikhonov and G. N. Sarkisov, to appear in:Zh. Fiz. Khim.
J. P. Hansen and I. R. McDonald,Theory of Simple Liquids, Academic Press, London (1986).
H. J. C. Berendsen, J. P. M. Postma, and W. F. Von Gunsteren, in:Intermolecular Forces, B. Pullman (ed.), Reidel, Dordrecht (1981).
J. O. Hirschfelder, C. F. Curtis, and R. B. Bird,Molecular Theory of Gases and Liquids, Wiley, New York (1954).
A. Ben-Naim and Y. Marcus,J. Chem. Phys.,81, No. 4, 2016–2027 (1984).
Additional information
Institute of Theoretical and Experimental Biophysics, Russian Academy of Sciences. Translated fromZhurnal Strukturnoi Khimii, Vol. 37, No. 4, pp. 736–741, July–August, 1996.
Translated by L. Smolina
Rights and permissions
About this article
Cite this article
Sarkisov, G.N., Tikhonov, D.A. Role of water structure in thermodynamics of nonpolar solvation. J Struct Chem 37, 633–638 (1996). https://doi.org/10.1007/BF02437177
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02437177