Abstract
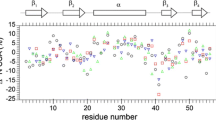
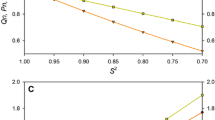
In proteins, dynamic mobility is an important feature of structure, stability, and biomolecular recognition. Uniquely sensitive to motion throughout the milli- to picosecond range, rates of transverse relaxation, R2, are commonly obtained for the characterization of chemical exchange, and the construction of motional models that attempt to separate overall and internal mobility. We have performed an in-depth study of transverse relaxation rates of backbone 15N nuclei in TNfn31−90, the third fibronectin type III domain from human tenascin. By combining the results of spin-echo (CPMG) and off-resonance T1ρ experiments, we present R2 rates at effective field strengths of 2 to 40 krad/s, obtaining a full spectrum of 16 independent R2 data points for most residues. Collecting such a large number of replicate measurements provides insight into intrinsic uncertainties. The median standard deviation in R2 for non-exchanging residues is 0.31, indicating that isolated measurements may not be sufficiently accurate for a precise interpretation of motional models. Chemical exchange events on a timescale of 570 μs were observed in a cluster of residues at the C terminus. Rates of exchange for five other residues were faster than the sampled range of frequencies and could not be determined. Averaged 'exchange free' transverse relaxation rates, R20, were used to calculate the diffusion tensor for rotational motion. Despite a highly asymmetric moment of inertia, the narrow angular dispersion of N-H vectors within the β sandwich proves insufficient to define deviations from isotropic rotation. Loop residues provide exclusive evidence for axially symmetric diffusion (Dpar/Dper=1.55).
Similar content being viewed by others
References
Akke, M., Liu, J., Cavanagh, J., Erickson, H.P. and Palmer, A.G. (1998) Nat. Struct. Biol., 5, 55–59.
Akke, M. and Palmer, A.G. (1996) J. Am. Chem. Soc., 118, 911–912.
Allerhand, A. and Gutowsky, H.S. (1965) J. Chem. Phys., 42, 1587–1599.
Arcus, V.A., Vuilleumier, S., Freund, S.M.V., Bycroft, M. and Fersht, A.R. (1995) J. Mol. Biol., 254, 305–321.
Banci, L., Bertini, I., Cavazza, C., Felli, I.C. and Koulougliotis, D. (1998) Biochemistry, 37, 12320–12330.
Bevington, P.R. and Robinson, D.K. (1992) Data Reduction and Error Analysis for the Physical Sciences (2nd ed.), McGraw-Hill, New York, NY.
Bloom, M., Reeves, L.W. and Wells, E.J. (1965) J. Chem. Phys., 42, 1615–1624.
Broadhurst, R.W., Hardman, C.H., Thomas, J.O. and Laue, E.D. (1995) Biochemistry, 34, 16608–16617.
Brüschweiler, R., Liao, X. and Wright, P.E. (1995) Science, 268, 886–888.
Carr, H.Y. and Purcell, E.M. (1954) Phys. Rev., 94, 630–638.
Clarke, J., Hamill, S.J. and Johnson, C.M. (1997) J. Mol. Biol., 270, 771–778.
Constantine, K.L., Friedrichs, M.S., Wittekind, M., Jamil, H., Chu, C.-H., Parker, R.A., Goldfarb, V., Mueller, L. and Farmer, B.T. (1998) Biochemistry, 37, 7965–7980.
Davis, D.G., Perlman, M.E. and London, R.E. (1994) J. Magn. Reson., B104, 266–275.
Deverell, C., Morgan, R.E. and Strange, J.H. (1970) Mol. Phys., 18, 552–559.
Farrow, N.A., Zhang, O., Szabo, D., Torchia, D.A. and Kay, L.E. (1995) J. Biomol. NMR, 6, 153–162.
Freund, S.M.V., Wong, K.-B. and Fersht, A.R. (1996) Proc. Natl. Acad. Sci. USA, 93, 10600–10603.
Gagné, S.M., Tsuda, S., Spyracopoulos, L., Kay, L.E. and Sykes, B.D. (1998) J. Mol. Biol., 278, 667–686.
Jin, D., Figueirido, F., Montelione, G.T. and Levy, R.M. (1997) J. Am. Chem. Soc., 119, 6923–6924.
Kay, L.E. (1998) Nat. Struct. Biol., 5, 513–517.
Kay, L.E., Keifer, P. and Saarinen, T. (1992) J. Am. Chem. Soc., 114, 10663–10665.
Kay, L.E., Torchia, D.A. and Bax, A. (1989) Biochemistry, 28, 8972–8979.
Kroenke, C.D., Loria, J.P., Lee, L.K., Rance, M. and Palmer, A.G. (1998) J. Am. Chem. Soc., 120, 7905–7915.
Lane, A.N. and Lefévre, J.-F. (1994) Methods Enzymol., 239, 596–619.
Leahy, D.J., Hendrickson, W.A., Aukhil, I. and Erickson, H.A. (1992) Science, 258, 987–991.
Lee, L.K., Rance, M., Chazin, W.J. and Palmer, A.G. (1997) J. Biomol. NMR, 9, 287–298.
Lipari, G. and Szabo, A. (1982a) J. Am. Chem. Soc., 104, 4546–4559.
Lipari, G. and Szabo, A. (1982b) J. Am. Chem. Soc., 104, 4559–4570.
Luginbühl, P., Pervushin, K.V., Iwai, H. and Wüthrich, K. (1997) Biochemistry, 36, 7305–7312.
Mandel, A.M., Akke, M. and Palmer, A.G. (1996) Biochemistry, 35, 16009–16023.
McIntosh, P.B., Frenkiel, T.A., Wollborn, U., McCormick, J.E., Klempnauer, K.H., Feeney, J. and Carr, M.D. (1998) Biochemistry, 37, 9619–9629.
Meekhof, A.E., Hamill, S.J., Arcus, V.L., Clarke, J. and Freund, S.M.V. (1998) J. Mol. Biol., 282, 181–194.
Mulder, F.A.A., de Graff, R.A., Kaptein, R. and Boelens, R. (1998) J. Magn. Reson., 131, 351–357.
Ogata, K., Kanei-Ishii, C., Sasaki, M., Hatanaka, H., Nagadoi, A., Enari, M., Nakamura, H., Nishimura, Y., Ishii, S. and Aarai, A. (1996) Nat. Struct. Biol., 3, 178–187.
Orekhov, V.Y., Pervushin, K.V. and Arseniev, A.S. (1994) Eur. J. Biochem., 219, 887–896.
Palmer, A.G. (1997) Curr. Opin. Struct. Biol., 7, 732–737.
Pervushin, K., Riek, R., Wider, G. and Wüthrich, K. (1997) Proc. Natl. Acad. Sci. USA, 94, 12366–12371.
Phan, I.Q.H., Boyd, J. and Campbell, I.A. (1996) J. Biomol. NMR, 8, 369–378.
Schurr, J.M., Babcock, H.P. and Fujimoto, B.S. (1994) J. Magn. Reson., B105, 211–224.
Szyperski, T., Luginbühl, P., Otting, G., Güntert, P. and Wüthrich, K. (1993) J. Biomol. NMR, 3, 151–164.
Tjandra, N., Feller, S.E., Pastor, R.W. and Bax, A. (1995) J. Am. Chem. Soc., 117, 12562–12566.
Tjandra, N., Wingfield, P., Stahl, S. and Bax, A. (1996) J. Biomol. NMR, 8, 273–284.
Vis, H., Vorgias, C.E., Wilson, K.S., Kaptein, R. and Boelens, R. (1998) J. Biomol. NMR, 11, 265–277.
Whittaker, S.B.M., Boetzel, R., MacDonald, C., Lian, L., Pommer, A.J., Reilly, A., James, R., Kleanthous, C. and Moore, G.R. (1998) J. Biomol. NMR, 12, 145–159.
Wong, K.-B., Freund, S.M.V. and Fersht, A.R. (1996) J. Mol. Biol., 259, 805–818.
Wyss, D., Dayie, K.T. and Wagner, G. (1997) Protein Sci., 6, 534–542.
Zinn-Justin, S., Berthault, P., Guenneugues, M. and Desvaux, H. (1997) J. Biomol. NMR, 10, 363–372.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meekhof, A.E., Freund, S.M. Separating the contributions to 15N transverse relaxation in a fibronectin type III domain. J Biomol NMR 14, 13–22 (1999). https://doi.org/10.1023/A:1008371332130
Issue Date:
DOI: https://doi.org/10.1023/A:1008371332130