Abstract
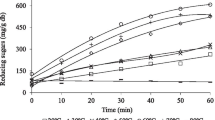
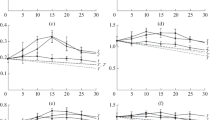
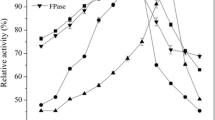
Immobilisation of trehalose phosphorylase from Schizophyllum commune by adsorption onto anion-exchange materials stabilised the enzyme activity at 30 °C by approx. 35-fold. Immobilised and free enzymes showed similar pH-dependence of activity but different inactivation behavior above 30 °C. A fixed-bed enzyme reactor produced α,α-trehalose at a stable substrate conversion of 80% with a productivity of 2.6 g l−1 h−1 for 72 h. Inhibition of trehalose phosphorylase by phosphate limited the productivity of a direct conversion of starch into α,α-trehalose.
Similar content being viewed by others
References
Crowe JH, Crowe LM, Chapman D (1984) Science 223: 701-703.
Eis C, Albert M, Dax K, Nidetzky, B (1998) FEBS Lett. 440: 440-443.
Elbein AD (1974) Adv. Carbohydr. Chem. Biochem. 30: 227-256.
Ikegami S, Kubota M, Sugimoto T, Miyake T (1995) European Patent 0 688 866 A1, Int. Cl. C12N 9/24.
Koh S, Shin H-J, Kim JS, Lee D-S, Lee SY (1998) Biotechnol. Lett. 20: 757-761.
Roser B (1991) Trends Food Sci. Technol. 2: 166-169.
Saito K, Kase T, Takahashi E, Takahashi E, Horinouchi S (1998) Appl. Environ. Microbiol. 64: 4340-4345.
Van Laere A (1989) FEMS Microbiol. Rev. 63: 201-210.
Weinhäusel A, Grießler R, Krebs A, Zipper P, Haltrich D, Kulbe KD, Nidetzky B (1997) Biochem. J. 326: 773-783.
Weinhäusel A, Nidetzky B, Blauensteiner B, Rohrbach B, Kulbe KD (1994) Appl. Microbiol. Biotechnol. 41: 510-516.
Yoshida M, Nakamura N, Horikoshi K (1998a) Enzyme Microb. Technol. 22: 71-75.
Yoshida M, Nakamura N, Horikoshi K (1998b) Starch/Stärke 49: 21-26.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klimacek, M., Eis, C. & Nidetzky, B. Continuous production of α,α-trehalose by immobilised fungal trehalose phosphorylase. Biotechnology Techniques 13, 243–248 (1999). https://doi.org/10.1023/A:1008986623007
Issue Date:
DOI: https://doi.org/10.1023/A:1008986623007