Summary
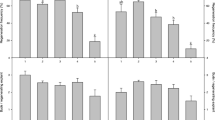
Callus induction, callus growth and organogenetic processes were investigated in hypocotyl and stem cultures of four species of Amaranthus each of which comprised several varieties. Callus formation occurred in almost all the varieties in 100% of explants. The combinations of naphtaleneacetic acid plus benzylaminopurine or 2,4-dichlorophenoxyacetic acid plus Kinetin were very effective in causing callus formation; the results for callus growth in presence of the same growth regulator combinations revealed in some cases significant differences among the varieties.
As far organogenesis-based on a few varieties-only A. caudatus and A. hypochondriacus responded well forming shoots from callus when cultured in presence of indoleacetic acid plus Kinetin and/or indoleacetic acid plus benzylaminopurine. Root regeneration was also observed in several varieties. Complete plantlets were obtained from rooted shoots.
The results are discussed in relation to hormonal effect and the genotype importance.
Similar content being viewed by others
References
Bagga, S., K., Venkateswarlu & S.K., Sopory, 1987. In vitro regeneration of plants from hypocotyl segments of Amaranthus paniculatus. Plant Cell Reports 6: 183–184.
Baroncelli, S., M., Buiatti, A., Bennici & M., Pagliai, 1974. Genetics of growth and differentiation in vitro of Brassica oleracea var. botrytis. III. Genetic correlations and ontogenetic unity. Z. Pflanzenzüchtg. 72: 275–282.
Buiatti, M., S., Baroncelli, A., Bennici, M., Pagliai & R., Tesi, 1974. Genetics of growth and differentiation in vitro of Brassica oleracea var. botrytis. II. An in vitro and in vivo analysis of a diallel cross. Z. Pflanzenzüchtg. 72: 269–274.
Evans, D.A., W.R. Sharp & C.E. Flick, 1981. Growth and behavior of cell culture: embryogenesis and organogenesis. p. 45–113. In: T.A. Thorpe (Ed). Plant Tissue Culture. Methods and application in agriculture. Academic Press.
Flores, H.E. & R.A., Teutonico, 1986. Amaranths (Amaranthus spp.): Potential grain and vegetable crops. p. 568–577. In: Y.P.S., Bajaj (Ed.) Biotechnology in Agriculture and Forestry, Vol. 2: Crops I. Springer-Verlag, Berlin, Heidelberg.
Flores, H.E., A., Thier & A.W., Galston, 1982. In vitro culture of grain and vegetable Amaranths (Amaranthus spp.). Amer. J. Bot. 69(7): 1049–1054.
Gamborg, O.L., J.P., Shyluk, D.S., Brar & F., Constabel, 1977. Morphogenesis and plant regeneration from callus of immature embryos of Sorghum. Plant Sci. Lett. 10: 67–74.
Gudu, S. & V.K., Gupta, 1988. Male sterility in the grain amaranth (Amaranthus hypochondriacus ex Nepal) variety Jumla. Euphytica 37: 23–26.
Lupi, M.C., A., Bennici & D., Gennai, 1985. In vitro culture of Bellevalia romana (L.) Rchb. I. Plant regeneration through adventitious shoots and somatic embryos. Protoplasma 125: 185–189.
Lupi, M.C., A., Bennici, F., Locci & D., Gennai, 1987. Plantlet formation from callus and shoot-tip culture of Helianthus annuus (L.). Plant Cell, Tissue and Organ Culture 11: 47–55.
Murashige, M. & F., Skoog, 1962. A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol. Plant. 15: 473–497.
Orton, T.J., 1980. Chromosomal variability in tissue cultures and regenerated plants of Hordeum. Theor. Appl. Genet. 56: 101–112.
Peters, I. & S., Jain, 1987. Geneties of grain amaranths. III. Gene-cytoplasmic male sterility. Heredity 78: 251–256.
Sears, R.G. & E.L., Deckard, 1982. Tissue culture variability in wheat callus induction and plant regeneration. Crop Sci. 22: 546–550.
Skoog, F. & C.O., Miller, 1957. Chemical regulation of growth and organ formation in plant tissue cultivated in vitro. In: Biological action of growth substances. 11th Symp. Soc. Exp. Biol. 11: 118–131.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bennici, A., Schiff, S. & Bovelli, R. In vitro culture of species and varieties of four Amaranthus L. species. Euphytica 62, 181–186 (1992). https://doi.org/10.1007/BF00041752
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00041752