Abstract
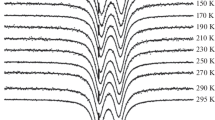
Mössbauer studies of the ubiquitous protein molecule ferritin and its synthetic “biomimic” polysaccharide iron complex (PIC) exhibit an anomaly in the Mössbauer spectrum wherein the recoil free fraction orf-factor has a sharp drop with respect to temperature as the temperature rises above 30 K for mammalian ferritin and 60 K for PIC. The anomaly coincides with the disappearance of hyperfine splitting, which is due to superparamagnetic relaxation above the blocking temperature. Different absorbers were used to experimentally investigate the effect of absorber thickness on the Mössbauer spectrum. The anomaly persists for thin absorbers. Also, spectra treated with FFT procedures to eliminate the thickness effect still exhibit this anomaly. Motion of the core with respect to the protein shell was also eliminated as a possible source for this phenomenon, by comparing the Debye temperature obtained from the temperature dependence of thef-factor and the isomer shift. A comparison of the magnetic anisotropy constants from magnetization studies with those obtained by relating the hyperfine fieldH of the Mössbauer spectra to the fluctuations of the magnetization imply that the ferritin and PIC molecules possess magnetic anisotropy energy which may not be strictly uniaxial. This, we believe, may be intimately connected with the mechanism causing thef-factor anomaly.
Similar content being viewed by others
References
J.Yariv et al., Biochem. J. 197 (1981) 171.
E.I. Stiefel and G.D. Watt, Nature 279 (1979) 81.
E.C. Theil, in:Advances in Inorganic Biochemistry, Vol. 5, eds. E.C. Theil, G.L. Eichorn and L.G. Marzilli (Elsevier, Amsterdam, 1983).
K.M. Towe and W.F. Bradley, J. Colloid, Interface Sci. 24 (1967) 384.
C.C. Ford et al., Phil. Trans. Roy. Soc. B 304 (1984) 551.
D.W. Rice et al. in:Advances in Inorganic Biochemistry, Vol. 5, eds. E.C. Theil, G.L. Eichorn and L.G. Marzilli (Elsevier, Amsterdam, 1983)
P.M. Harrison, in:Advances in Inorganic Biochemistry, Vol. 5, eds. E.C. Theil, G.L. Eichorn and L.G. Marzilli (Elsevier, Amsterdam, 1983).
D.M. Lawson et al., Nature 349 (1991) 542.
D.M. Lawson et al., FEBS Lett. 254 (1989) 207.
E.R. Bauminger et al., Biochem. Biophys. Acta 1118 (1991) 48.
D.L. Jacobs et al., Biochemistry 28 (1989) 1650.
A. Treffy et al., FEBS Lett. 302 (1992) 108.
A. Blaise et al., Compt. Rend. Acad. Sci. Paris 261 (1965) 2310; 265 (1967) 1077.
R.B. Frankel et al., Hyp. Int. 33 (1987) 233.
B. Kolk, in:Dynamical Properties of Solids, Vol. 5, eds. G.K. Horton and A.A. Maradudin (North-Holland, Amsterdam, 1984)
H. Frauenfelder,The Mössbauer Effect (Benjamin, New York, 1963).
J.F. Sanders, Mich. Med. 7 (1968) 726
R.W. Nexton et al., Clin. Trials J. 17 (1980) 106; O.W. Crawford, Ill. Med. J. (1970).
K.A. Berg et. al., J. Inorg. Biochem. 22 (1984) 125.
M-E.Y. Mohie-Eldin, PhD Thesis, Tufts University, Medford, MA, USA (1992).
U.S. Patent No. 3821192 process for preparing an iron-saccharide complex.
T.G. Spiro and P. Saltman, Struct. Bonding 6 (1969) 116
P.J. Murphy et al., J. Colloid Interf. Sci. 56 (1976) 312.
M-E.Y. Mohie-Eldin, R.B. Frankel and L. Gunther, J. Magn. Magn. Mater. 135 (1994) 65.
F. Varret, J. Phys. Chem. Solids 37 (1976) 265.
N.N. Greenwood and T.C. Gibb,Mössbauer Spectroscopy (Chapman and Hall, London, 1971).
V.I. Goldanskii and R.H. Herber,Chemical Applications of Mössbauer Spectroscopy (Academic Press, New York, 1968).
J. Herberle and S. Franco, Z. Naturforsch. 10 (1968) 1439.
S. Margulies and J.R. Ehrman, Nucl. Instr. Meth. 12 (1961) 131.
S.L. Ruby and J.M. Hicks, Rev. Sci. Instr. 33 (1962) 27.
M.C.D. Ure and P.A. Flinn, in:Mössbauer Effect Methodology, Vol. 7, ed. I.J. Gruverman (Plenum Press, New York, 1971) p. 245.
R.B. Frankel et al., Hyp. Int. 66 (1991) 71.
P. Roggwiller and W. Kundig, Solid State Commun. 12 (1973) 901.
S. Mørup and H. Topsøe, Appl. Phys. 11 (1976) 63.
S. Mørup et al., J. Phys. (Paris) 37 (1976) C6–287.
S. Mørup, Hyp. Int. 60 (1990) 959
S. Mørup, J. Magn. Magn. Mater. 37 (1983) 39.
J.M. Williams et al., Phys. Med. Biol. 23 (1978) 835.
M. Abramowitz and I.A. Stegun, eds.,Handbook of Mathematical Functions (Dover, New York, 1972) pp. 297, 305, 319.
T.G. St. Pierre et al., J. Magn. Mater. 69 (1987) 276.
M.P.A. Viegers and J.M. Trooster, Phys. Rev. B 15 (1977) 72.
I.P. Suzdalev et al., Sov. Phys. JETP 24 (1968) 79.
M.Hayashi et al., J.Phys.C13 (1980) 681.
A.M. van der Kraan, Phys. Stat. Sol. 18a (1973) 215.
J.W. Niemantsverdriet et al., Phys. Lett. 100A 8 (1984) 445.
J. Heberle, Möss. Effect. Meth. 7 (1971) 299.
K. A. Hardy et al., Nucl. Instr. Meth. 86 (1970) 171.
M-E.Y. Mohie-Eldin and L. Gunther, in:Proc. Int. Workshop on Studies of Magnetic Properties of Fine Particles and Their Relevance to Materials Science, Rome, eds. J.L. Dormann and D. Fiorani (Elsevier, Amsterdam, 1992) p.403.
M-E.Y. Mohie-Eldin and L. Gunther, J. Magn. Magn. Mater. 127 (1993) 346.
L. Gunther and M.-E.Y. Mohie-Eldin, J. Magn. Magn. Mater. 129 (1994) 334.
C.Hawkins, private communication.
S.H. Bell, Biochem. Biophys. Acta. 787 (1984) 227
E.R. Bauminger, Biochem. Biophys. Acta. 623 (1980) 237.
Author information
Authors and Affiliations
Additional information
This work was carried out in partial fulfilment of the requirements of the PhD degree at Tufts University.
Rights and permissions
About this article
Cite this article
Mohie-Eldin, M.E.Y., Frankel, R.B., Gunther, L. et al. The anomalous Mössbauer fraction of ferritin and polysaccharide iron complex (PIC). Hyperfine Interact 96, 111–138 (1995). https://doi.org/10.1007/BF02066277
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02066277