Summary
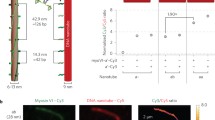
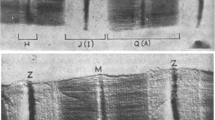
Exchange of myosin molecules between synthetic thick filaments was examined by fluorescence energy transfer and visualized by electron microscopy using streptavidin-gold to detect exchanged biotinylated myosin molecules. N-hydroxysuccinimidobiotin (NHS-biotin) was covalently linked to purified adult chicken pectoralis myosin to obtain assembly-competent biotinylated myosin molecules. Two distinct classes of synthetic filaments, distinguishable by length, were prepared. Biotinylated filaments (575±100 nm) were assembled by a quick dilution (QD) method and unlabelled filaments (1025±250 nm) were obtained by a sequential dilution (SD). The two filament population maintained their distinct length distributions even when mixed. To measure exchange, biotinylated short (QD) filaments were combined with unlabelled long (SD) filaments at a 1∶5 ratio, sampled at varying times and the entry of biotinylated myosin into the previously unlabelled long filaments visualized by the addition of streptavidin-gold. The number of gold particles per micron was examined for fully biotinylated short filaments (<700 nm), unlabelled long filaments (>900 nm), and exchanged filaments. Equivalent binding of streptavidin-gold to the two filament types was detected by 60 min suggesting randomization of biotinylated monomers by this time. The precise location of streptavidin-gold sites on the long filaments was also measured. Although labeling was detected along the full length of the filaments, at the earliest time points (5 min) filament ends contained twice the number of gold particles as the filament centers. Approximately equivalent labeling along the entire length of the filaments was observed by 60 min. These results provide additional support for our earlier report of extensive myosin exchange between synthetic thick filaments and show that extensive exchange takes place rapidly along the full length of synthetic thick filaments.
Similar content being viewed by others
References
Bader, D., Masaki, T. &Fischman, D. A. (1982) Immunochemical analysis of myosin heavy chain during avian myogenesisin vivo andin vitro.J. Cell Biol. 95, 763–70.
Bahler, M., Eppenberger, H. M. &Wallimann, T. (1985a) Novel thick filament protein of chicken pectoralis muscle: the 86 kd protein. I. Purification and characterization.J. Mol. Biol. 186, 381–91.
Bahler, M., Eppenberger, H. M. &Wallimann, T. (1985b) Novel thick filament protein of chicken pectoralis muscle: the 86 kd protein. II. Distribution and localization.J. Mol. Biol. 186, 393–401.
Gauthier, G. F. (1990) Differential distribution of myosin isoforms among the myofibrils of individual developing muscle fibers.J. Cell. Biol. 110, 693–701.
Godfrey, J. E. &Harrington, W. F. (1970) Self-association in the myosin system at high ionic strength. I. Sensitivity of the interaction to pH and ionic environment.Biochemistry 9, 886–93.
Green, N. M. (1975). Avidin.Advances in Protein Chemistry 29, 85–133.
Grove, B. K., Kurer, V., Lehner, C., Doetschman, T. C., Perriard, J.-C. &Eppenberger, H. M. (1984) A new 185.000-dalton skeletal muscle protein detected by monoclonal antibodies.J. Cell Biol. 98, 518–24.
Horisberger, M. &Rosett, J. (1977) Colloidal gold, a useful marker for transmission and scanning electron microscopy.J. Histochem. Cytochem. 25, 295–305.
Huxley, H. E. (1963) Electron microscope studies on structure of natural and synthetic protein filaments from striated muscle.J. Molec. Biol. 7, 281–308.
Johnson, C. S., Mckenna, N. M. &Wang, Y. (1988) Association of microinjected myosin and its subfragments with myofibrils in living muscle cells.J. Cell Biol. 107, 2213–21.
Josephs, R. &Harrington, W. F. (1968) On the stability of myosin filaments.Biochemistry 7, 2834–47.
Katsura, I. &Noda, H. (1971) Studies on the formation and physical chemical properties of synthetic myosin filaments.J. Biochem. 69, 219–29.
Kreis, T. E., Geiger, B. &Schlessinger, J. (1982) Mobility of microinjected rhodamine actin within living chicken gizzard cells determined by fluorescence photobleaching recovery.Cell 29, 835–45.
Kristofferson, D., Mitchison, T. &Kirschner, M. (1986) Direct observation of steady-state microtubule dynamics.J. Cell Biol. 102, 1007–19.
Masaki, T. &Takaiti, O. (1974) M-protein.J. Biochem. (Tokyo) 75, 367–80.
Maw, M. C. &Rowe, A. J. (1980) Fraying of A-filaments into three sub-filaments.Nature 286, 412–14.
Maruyama, K., Kimura, S., Ohashi, K. &Kuwano, Y. (1981) Connectin, and elastic protein of muscle. Identification of titin with connectin.J. Biochem. (Tokyo)89, 701–9.
Mckenna, N. M., Meigs, J. B. &Wang, Y. (1985) Exchangeability of alpha-actinin in living cardiac fibroblasts and muscle cells.J. Cell Biol. 101, 2223–32.
Mittal, B., Sanger, J. M. &Sanger, J. W. (1987) Visualization of myosin in living cells.J. Cell Biol. 105, 1753–60.
Offer, G., Moos, C. S. &Starr, R. (1973) A new protein of the thick filaments of vertebrate skeletal myofibrils. Extraction, purification and characterization.J. Mol. Biol. 74, 653–76.
Pardee, J. D., Simpson, P. A., Stryer, L. &Spudich, J. A. (1982) Actin filaments undergo limited subunit exchange in physiological salt conditions.J. Cell Biol. 94, 316–24.
Pepe, F. A. (1983) Macromolecular assembly of myosin. In:Muscle and Nonmuscle Motility. (edited byStracher, A.) pp. 105–149. New York: Academic Press.
Pepe, F. A., Drucker, B. &Chowrashi, P. K. (1986) The myosin filament, XI: filament assembly.Prep. Biochem. 16, 99–132.
Price, M. (1987) Skelemins: cytoskeletal proteins located at the periphery of M-discs in mammalian striated muscle.J. Cell Biol. 104, 1325–36.
Saad, A. D., Fischman, D. A. &Pardee, J. D. (1986a) Fluorescence energy transfer studies of myosin thick filament assembly.Biophys. J. 49, 140–2.
Saad, A. D., Mathews, A. P., Tan, I. P. &Sorrentino, A. M. (1990) Myosin thick filament stability in the absence and presence of C-protein.Anatomical Record (in Press).
Saad, A. D., Pardee, J. D. &Fischman, D. A. (1986b) Dymanic exchange of myosin molecules between thick filaments.Proc. Natl. Acad. Sci. (USA)83, 9483–7.
Salmon, E. D., Leslie, R. J., Saxton, W. M., Krow, M. L. &Mcintosh, J. R. (1984) Spindle microtubule dynamics in sea urchin embryos: analysis using a fluorescein-labeled tubulin and measurements of fluorescence redistribution after laser photobleaching.J. Cell Biol. 99, 2165–74.
Shimuzu, T., Dennis, J. E., Masaki, T. &Fischman, D. A. (1985) Axial arrangement of the myosin rod in vertebrate thick filaments: immunoelectron microscopy with a monoclonal antibody to light meromyosin.J. Cell Biol. 101, 1115–23.
Sutoh, K., Yamamoto, K. &Wakabayashi, T. (1984) Electron microscopic visualization of the SH1 thiol of myosin by the use of an avidin-biotin system.J. Molec. Bio. 178, 323–39.
Trybus, K. M. &Lowey, S. (1987) Subunit exchange between smooth muscle myosin filaments.J. Cell Biol. 105, 3021–30.
Tyler, J. M. &Branton, D. (1980) Rotary shadowing of extended molecules dried from glycerol.J. Ultrastruct. Res. 71, 95–102.
Wallimann, T., Turner, D. C. &Eppenberger, H. M. (1977) Localization of creatine kinase isozymes in myofibrils.J. Cell Biol. 75, 297–317.
Wang, D., McClure, J. &Tu, A. (1979) Titin: major myofibrillar components of striated muscle.Proc. Natl. Acad. Sci. (USA)76, 3698–702.
Wenderoth, M. P. &Eisenberg, B. R. (1987) Incorporation of nascent myosin heavy chains into thick filaments of cardiac myocytes in thyroid treated rabbits.J. Cell Biol. 105, 2771–80.
Wrigley, N. G. (1968) The lattice spacing of crystalline catalase as an internal standard of length in electron microscopy.J. Ultrastruct. Res. 24, 454–64.
Zak, R., Martin, A. F., Prior, G. &Rabinowitz, M. (1977) Comparison of turnover of several myofibrillar proteins and critical evaluation of double isotope method.J. Cell Biol. 252, 3430–5.
Zak, R., Martin, A. F. &Blough, R. (1979) Assessment of protein turnover by use of radioisotopic tracers.Physiological Rev. 59, 407–47.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saad, A.D., Dennis, J.E., Tan, I.P. et al. Visualization of myosin exchange between synthetic thick filaments. J Muscle Res Cell Motil 12, 225–234 (1991). https://doi.org/10.1007/BF01745111
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01745111