Abstract
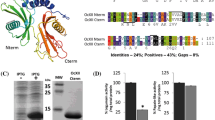
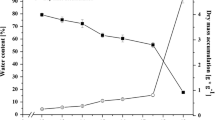
Families of papain- and legumain-like cysteine proteinases (CPR) were found in Vicia seeds. cDNAs and antibodies were used to follow organ specificity and the developmental course of CPR-specific mRNAs and polypeptides. Four papain-like cysteine proteinases (CPR1, CPR2, proteinase A and CPR4) from vetch seeds (Vicia sativa L.) were analysed. CPR2 and its mRNA were already found in dry embryonic axes. CPR1 was only detected there during early germination. Both CPR1 and CPR2 strongly increased later during germination. In cotyledons, both CPR1 and CPR2 were only observed one to two days later than in the axis. Proteinase A was not found in axes. In cotyledons it could only be detected several days after seeds had germinated. CPR4 mRNA and polypeptide were already present in embryonic axes and cotyledons during seed maturation and decreased in both organs during germination. Purified CPR1, CPR2 and proteinase A exhibited partially different patterns of globulin degradation products in vitro. Although the cDNA-deduced amino acid sequence of the precursor of proteinase A has an N-terminal signal peptide, the enzyme was not found in vacuoles whereas the other papain-like CPRs showed vacuolar localization. Four different legumain-like cysteine proteinases (VsPB2, proteinase B, VnPB1 and VnPB2) of Vicia species were analysed. Proteinase B and VnPB1 mRNAs were detected in cotyledons and seedling organs after seeds had germinated. Proteinase B degraded globulins isolated from mature vetch seeds in vitro. VsPB2 and proteinase B are localized to protein bodies of maturing seeds and seedlings, respectively, of V. sativa. Like VsPB2 from V. sativa, also VnPB2 of V. narbonensis corresponds to vacuolar processing enzymes (βVPE). Based on these results different functions in molecular maturation and mobilization of storage proteins could be attributed to the various members of the CPR families.
Similar content being viewed by others
References
Alonso, J.M. and Granell, A. 1995. A putative vacuolar processing protease is regulated by ethylene and also during fruit ripening in Citrus fruit. Plant Physiol. 109: 541–547.
Barrett, A.J., Rawlings, N.D. and Woessner, J.F. 1998. Handbook of Proteolytic Enzymes, Academic Press, San Diego, CA.
Baumgartner, B. and Chrispeels, M.J. 1977. Purification and characterization of vicilin peptidohydrolase, the major endopeptidase in the cotyledons of mung-bean seedlings. Eur. J. Biochem. 77: 223–233.
Becker, C., Fischer, J., Nong, V.H. and Müntz, K. 1994. PCR cloning and expression analysis of cDNAs encoding cysteine proteinases from germinating seeds of Vicia sativa. Plant Mol. Biol. 26: 1207–1212.
Becker, C., Shutov, A.D., Nong, V.H., Senyuk, V.I., Jung, R., Horstmann, C., Fischer, J., Nielsen, N.C. and Müntz, K. 1995. Purification, cDNA cloning and characterization of proteinase B, an asparagine-specific endopeptidase from germinating vetch (Vicia sativa L.) seeds. Eur. J. Biochem. 228: 456–462.
Becker, C., Senyuk, V.I., Shutov, A.D., Nong, V.H., Fischer, J., Horstmann, C. and Müntz, K. 1997. Proteinase A, a storageglobulin-degrading endopeptidase of vetch (Vicia sativa L.) seeds, is not involved in early steps of storage protein mobilization. Eur. J. Biochem. 248: 304–312.
Bewley, J.D. and Black, M. 1994. Seeds: Physiology of Development and Germination, 2nd ed., Plenum, New York/London.
Borisjuk, L., Weber, H., Panitz, R., Manteuffel, R. and Wobus, U. 1995. Embryogenesis of Vicia faba L.: histodifferentiation in relation to starch and storage protein biosynthesis. J. Plant Physiol. 147: 203–218.
DeBarros, E.G. and Larkins, B.A. 1990. Purification and characterization of zein-degrading proteases from endosperm of germinating maize seeds. Plant Physiol. 94: 297–303.
Fischer, J. 1996. Charakterisierung einer Cysteinproteinase und ihrer cDNA aus Keimlingen der Saatwicke (Vicia sativa L.). Dissertation, Mathematisch-Naturwissenschaftliche Fakultät, Universität Halle, Germany.
Halfon, S., Patel, S., Vega, F., Zurawski, S. and Zurawski, G. 1998. Autocatalytic activation of human legumain at aspartic acid residues. FEBS Lett. 438: 114–118.
Hara-Nishimura, I., Inoue, K. and Nishimura, M. 1991. A unique vacuolar processing enzyme responsible for conversion of serveral proprotein precursors into mature forms. FEBS Lett. 294: 89–93.
Hara-Nishimura, I., Takeuchi, Y. and Nishimura, M. 1993. Molecular characterization of a vacuolar processing enzyme related to a putative cysteine proteinase of Schistosoma mansoni. Plant Cell 5: 1651–1659.
Hara-Nishimura, I., Kinoshita, T., Hiraiwa, N. and Nishimura, M. 1998. Vacuolar processing enzymes in protein storage vacuoles and lytic vacuoles. J. Plant Physiol. 152: 668–674.
Heim, U., Bäumlein, H. and Wobus, U. 1994. The legumin gene family: a reconstructed Vicia faba legumin gene encoding a highmolecular-weight subunit related to type B genes. Plant Mol. Biol. 25: 131–135.
Hiraiwa, N., Takeuchi, Y., Nishimura, M. and Hara-Nishimura, I. 1993. A vacuolar processing enzyme in maturing and germinating seeds: its distribution and associated changes during development. Plant Cell Physiol. 34: 1197–1204.
Hiraiwa, N., Nishimura, M. and Hara-Nishimura, I. 1999. Vacuolar processing enzyme is self-catalytically activated by sequential removal of the C-terminal and N-terminal propeptides. FEBS Lett. 447: 213–216.
Ishii, S. 1994. Legumain: asparaginyl endopeptidases. Meth. Enzymol. 244: 604–615.
Jones, J.T. and Mullet, J.E. 1995. A salt-and dehydration-inducible pea gene, Cyp15a, encodes a cell-wall protein with sequence similarity to cysteine proteinases. Plant Mol. Biol. 28: 1055–1065.
Jung, R., Scott, M.P., Nam, Y.-W., Beaman, T.W., Bassüner, R., Saalbach, I., Müntz, K. and Nielsen, N.C. 1998. The role of proteolysis in the processing and assembly of 11S seed globulins. Plant Cell 10: 343–357.
Kinoshita, T., Nishimura, M. and Hara Nishimura, I. 1995a. Homologues of a vacuolar processing enzyme that are expressed in different organs in Arabidopsis thaliana. Plant Mol. Biol. 29: 81–89.
Kinoshita, T., Nishimura, M. and Hara-Nishimura, I. 1995b. The sequence and expression of the y VPE gene, one member of a family of three genes for vacuolar processing enzymes in Arabidopsis thaliana. Plant Cell Physiol. 36: 1555–1562.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lahn, D.N., Shutov, A.D. and Vaintraub, I.A. 1985. Hydrolysis of vetch legumin by endogenous proteinases. Biokhimija (Russian) 50: 1076–1081.
Lawrence, M.C., Izard, T., Beuchat, M., Blagrove, R.J. and Colman, P.M. 1994. Structure of phaseolin at 2.2 Å resolution. Implication for a common vicilin/legumin structure and the genetic engineering of storage proteins. J. Mol. Biol. 238: 748–776.
Linnestad, C., Doan, D.N.P., Brown, R.C., Lemmon, B.E., Meyer, D.J., Jung, R. and Olsen, A.-O. 1998. Nucellain, a barley homologue of the dicot vacuolar processing protease, is localized in nucellar cell walls. Plant Physiol. 118: 1169–1180.
Mitsuhashi, W. and Minamikawa, T. 1989. Synthesis and posttranslational activation of sulfhydryl-endopeptidase in cotyledons of germinating Vigna mungo seeds. Plant Physiol. 89: 274–279.
Müntz, K. 1996. Proteases and proteolytic cleavage of storage proteins in developing and germinating dicotyledonous seeds. J. Exp. Bot. 47: 605–622.
Müntz, K., Becker, C., Pancke, J., Schlereth, A., Fischer, J., Horstmann, C., Kirkin, V., Neubohn, B., Senyuk, V.I. and Shutov, A.D. 1998. Protein degradation and nitrogen supply during germination and seedling growth of vetch (Vicia sativa L.). J. Plant Physiol. 152: 683–691.
Nong, V.H., Becker, C. and Müntz, K. 1995. cDNA cloning for a putative cysteine proteinase from developing seeds of soybean. Biochim. Biophys. Acta 1261: 435–438.
Okamoto, T. and Minamikawa, T. 1994. Post-translational processing of a carboxy-terminal propeptide containing a KDEL sequence of plant vacuolar cysteine endopeptidase (SH-EP). FEBS Lett. 351: 31–34.
Okamoto, T. and Minamikawa, T. 1995. Purification of a processing enzyme (VmPE-1) that is involved in post-translational processing of a plant cysteine endopeptidase (SH-EP). Eur. J. Biochem. 231: 300–305.
Okamoto, T. and Minamikawa, T. 1998. A vacuolar cysteine endopeptidase (SH-EP) that digests seed storage globulin: characterization, regulation of gene expression and post-translational processing. J. Plant Physiol. 152: 675–682.
Okamoto, T. and Minamikawa, T. 1999. Molecular cloning and characterization of Vigna mungo processing enzyme 1 (VmPE-1), an asparaginyl endopeptidase involved in post-translational processing of a vacuolar endopeptidase (SH-EP). Plant Mol. Biol. 39: 63–73.
Panitz, R., Borisjuk, L., Manteuffel, R. and Wobus, U. 1995. Transient expression of storage-protein genes during early embryogenesis of Vicia faba: synthesis and metabolization of vicilin and legumin in the embryo, suspensor and endosperm. Planta 196: 765–774.
Panitz, R., Manteuffel, R. and Wobus, U. 1999. Tobacco embryogenesis: storage protein accumulating cells of embryo, suspensor and endosperm are able to undergo cytokinesis. Protoplasma 207: 31–42.
Rawlings, N.D. and Barrett, A.J. 1994. Families of cysteine peptidases. Meth. Enzymol. 244: 461–485.
Rotari, V.I., Senyuk, V.I., Jivotovskaya, A.V., Horstmann, C. and Vaintraub, I.A. 1997. Proteinase A-like enzyme from germinated kidney bean seeds. Its action on phaseolin and vicilin. Physiol. Plant. 100: 171–177.
Saalbach, G., Christov, V., Jung, R., Saalbach, I., Manteuffel, R., Kunze, G., Brambarov, K. and Müntz, K. 1995. Stable expression of vicilin from Vicia faba with eight additional single methionine residues but failure of accumulation of legumin with an attached peptide segment in tobacco seeds. Mol. Breed. 1: 245–258.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
Schlereth, A., Becker, C., Horstmann, C. and Müntz, K. 2000. Comparison of globulin mobilization and cysteine proteinases in embryonic axes and cotyledons during germination and seedling growth of vetch (Vicia sativa L.) seeds. J. Exp. Bot., in press.
Schmid, M., Simpson, D. and Gietl, C. 1999. Programmed cell death in castor bean endosperm is associated with the accumulation and release of a cysteine endoeptidase from ricinosomes. Proc. Natl. Acad. Sci USA 96: 14159–14164.
Schmid, M., Simpson, D., Kalousek and Gietl, C. 1998. A cysteine endopeptidase with a C-terminal KDEL motif isolated from castor bean endosperm is a marker enzyme for the ricinosome, a putative lytic compartment. Planta 206: 466–475.
Scott, M.P., Jung, R., Müntz, K. and Nielsen, N.C. 1992. A protease responsible for post-translational cleavage of a conserved Asn-Gly linkage in glycinin, the major seed storage protein of soybean. Proc. Natl. Acad. Sci. USA 89: 658–662.
Senyuk, V.I., Rotari, V., Becker, C., Zakharov, A., Horstmann, C., Müntz, K. and Vaintraub, I. 1998. Does an asparaginylspecific cysteine endopeptidase trigger phaseolin degradation in cotyledons of kidney bean seedlings? Eur. J. Biochem. 258: 546–558.
Shimada, T., Niraiwa, N., Hishimura, M. and Hara-Nishimura, I. 1994. Vacuolar processing enzyme of soybean that converts proproteins to the corresponding mature forms. Plant Cell Physiol. 35: 713–718.
Shutov, A.D. and Vaintraub, I.A. 1987. Degradation of storage proteins in germinating seeds. Phytochemistry 26: 1557–1566.
Shutov, A.D., Lahn, D.N. and Vaintraub, I.A. 1982. Purification and partial characterization of proteinase B from germinating vetch seeds. Biokhimija (Russian) 47: 814–821.
Steffens, P., Nong, V.H., Hillmer, S., Saalbach, I. and Müntz, K. 1997. Subcellular localization of the 2S globulin narbonin in seeds of Vicia narbonensis. Planta 203: 44–50.
Takeda, O., Miura, Y., Mitta, M., Matsushita, H., Kato, I., Abe, Y., Yokosawa, H. and Ishii, S. 1994. Isolation and analysis of cDNA encoding a precursor of Canavalia ensiformis asparaginyl endopeptidase (legumain). J. Biochem. 116: 541–546.
Tiedemann, J., Neubohn, B. and Müntz, K. 2000. Different functions of vicilin and legumin are reflected in the histopattern of globulin mobilization during germination of vetch (Vicia sativa L.). Planta, in press.
Wilson, K.A. 1986. Role of proteolytic enzymes in the mobilization of protein reserves in germinating dicot seeds. In: M.J. Dalling (Ed.), Plant Proteolytic Enzymes, CRC Press, Boca Raton, FL, pp. 19–47.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fischer, J., Becker, C., Hillmer, S. et al. The families of papain- and legumain-like cysteine proteinases from embryonic axes and cotyledons of Vicia seeds: developmental patterns, intracellular localization and functions in globulin proteolysis. Plant Mol Biol 43, 83–101 (2000). https://doi.org/10.1023/A:1006456615373
Issue Date:
DOI: https://doi.org/10.1023/A:1006456615373