Summary
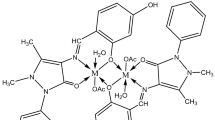
The interaction of 1-benzoin-4-phenylthiosemicarbazone (H2 BPS) with some transition metal ions has been investigated. The ligand can function as a tridentate chelating agent, giving M(HBPS)2 and M(BPS). Potentiometric studies proved that the mechanism of chelation is based on hydrogen ion liberation. Spectral studies in solution show that the ligand could be used for the microdetermination of CuIIions. On the basis of magnetic and spectral data, an octahedral structure is proposed for the CoII and NiII complexes and a square-planar structure for the CuII complex. The corrosion inhibition of aluminium in Cl3CCO2H using H2BPS is studied. The electrical conductivity of H2BPS and of its complexes have been measured. The ligand shows an activation energy in the range of semiconducting materials. The antimicrobial activity of all compounds has also been demonstrated.
Similar content being viewed by others
References
K. H. Reddy and D. V. Reddy,Ind. J. Chem.,22, 73 (1983).
K. N. Thimmaiah, H. S. Gowda and M. Ahmed,Ind. J. Chem.,22, 690 (1983).
M. I. Khalifa, K. M. Ibrahim and A. A. El-Asmy,Ind. J. Chem., in press.
M. J. M. Campbel,Coord. Chem. Rev.,15, 279 (1975).
N. P. Bauu-Hoi, T. B. Voc and N. D. Xuong,Bull. Soc. Chim. France, 694 (1955).
J. P. Scovill, D. L. Klayman and C. E. Franchino,J. Med. Chem.,25, 1261 (1982).
M. M. Mostafa, A. A. El-Asmy and G. M. Ibrahim,Transition Met. Chem.,8, 54 (1983).
A. A. El-Asmy, M. A. Morsi and A. A. El-Shafei,Transition Met. Chem.,11, 494 (1986).
W. J. Geary,Coord. Chem. Rev.,7, 81 (1971).
W. C. Vosburgh and G. R. Cooper,J. Am. Chem. Soc.,63, 937 (1941).
K. M. Purohit and D. V. Ramana,Ind. J. Chem.,22, 520 (1983).
C. K. Jorgenson,J. Inorg. Nucl. Chem.,24, 1521 (1962).
M. E. Khalifa, A. A. El-Asmy, M. M. Bekheit and M. M. Mostafa,Synth. React. Inorg. Met-Org. Chem.,16, 1305 (1986).
K. M. Ibrahim, A. A. El-Asmy, M. M. Bekheit and M. M. Mostafa,Transition Met. Chem.,10, 175 (1985).
M. Karo, H. B. Jonassen and J. C. Fanning,Chem. Revs.,64, 99 (1964).
L. Sacconi and M. Giampolini,J. Chem. Soc., A, 273 (1974).
A. A. El-Asmy and M. Mounir,Transition Met. Chem., in press.
A. A. El-Asmy, A. S. Babaqi and A. A. Al-Hubishi,Transition Met. Chem., in press.
A. A. El-Asmy, Y. M. Shaibi, I. M. Shedaiwa and M. A. Khatab,Synth. React. Inorg. Met.-Org. Chem., in press.
A. I. Vogel,A Text Book of Quantitative Inorganic Analysis, Longman, London (1961).
J. Lewis and R. G. Wilkins,Modern Coordination Chemistry, Interscience, New York (1960).
A. A. Ashour, M. N. El-Gharib, E. Kandeel and E. B. Moawad,Tanta, Med. J. Supp. I,11, 59 (1983).
A. A. Abou-Zeid and Y. M. Shehata,Ind. J. Pharmacy,31, 72 (1969).
American Society for Testing and Materials,Annual book of ASTM standards, 10 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Asmy, A.A., Shaibi, Y.M., Babaqi, A.S. et al. Physical, chemical and biological studies on transition metal complexes of chelating tridentate ligand. Transition Met Chem 13, 332–335 (1988). https://doi.org/10.1007/BF01225121
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01225121