Abstract
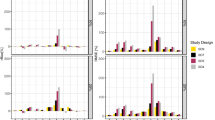
In a simulation study of the estimation of population pharmacokinetic parameters, including fixed and random effects, the estimates and confidence intervals produced by NONMEM were evaluated. Data were simulated according to a monoexponential model with a wide range of design and statistical parameters, under both steady state (SS) and nonSS conditions. Within the range of values for population parameters commonly encountered in research and clinical settings, NONMEM produced parameter estimates for CL, V, σCL,and σe which exhibit relatively small biases. As the range of variability increases, these biases became larger and more variable. An important exception was bias in the estimate for σv which was large even when the underlying variability was small. NONMEM standard error estimates are appropriate as estimates of standard deviation when the underlying variability is small. Except in the case of CL,standard error estimates tend to deteriorate as underlying variability increases. An examination of confidence interval coverage indicates that caution should be exercised when the usual 95% confidence intervals are used for hypothesis testing. Finally, simulationbased corrections of point and interval estimates are possible but corrections must be performed on a casebycase basis.
Similar content being viewed by others
References
J. L. Steimer, A. Mallet, and F. Mentre. Estimating interindividual pharmacokinetic variability.Variability in Drug Therapy: Description, Estimation and Control, In M., Rowland, L. B. Sheiner, and J.-L. Steimer (eds.), Raven Press, New York, 1985, pp. 65–111.
S. L. Beal and L. B. Sheiner. The NONMEM system.Am. Statist. 34:118–119 (1980).
L. B. Sheiner, B. Rosenberg, and V. V. Marathe. Estimation of population characteristics of pharmacokinetic parameters from routine clinical data.J. Pharmacokin. Biopharm. 5:445–479 (1977).
T. H. Grasela, L. B. Sheiner, B. Rambeck, H. E. Boenigk, A. Dunlop, P. W. Mullen, J. Wadsworth, A. Richens, T. Ishazaki, K. Chiba, H. Miura, K. Minagawa, P. G. Blain, J. C. Mucklow, C. T. Bacon, and M. Rawlins. Steady-state pharmacokinetics of phenytoin from routinely collected patient data.Clin. Pharmacokin. 8:355–364 (1983).
T. H. Grasela and L. B. Sheiner. Population pharmacokinetics of procainamide from routine clinical data.Clin. Pharmacokin. 9:545–554 (1984).
S. Vozeh, G. Katz, V. Steiner, and F. Follath. Population pharmacokinetic parameters in patients treated with oral mexilitene.Eur. J. Clin. Pharmacol. 23:445–451 (1982).
S. Vozeh, M. Wenk, and F. Follath. Experience with NONMEM: Analysis of serum concentration data in patients treated with tnexilitene and lidocaine.Drug Metab. Rev. 15:305–315 (1984).
A. W. Kelman, A. H. Thompson, B. Whiting, S. M. Bryson, D. A. Streedman, G. E. Mawer, and L. A. Samba-Donga. Estimation of gentamicin clearance and volume of distribution in neonates and young children.Br. J. Clin. Pharmacol. 18:685–692 (1984).
D. R. Mungall, T. M. Ludden, S. Marshall, D. W. Hawkins, R. L. Talbert, and M. H. Crawford. Population pharmacokinetics of racemic warfarin in aduit patients.J. Pharmacokin. Biopharm. 13:213–227 (1985).
T. H. Grasela and S. M. Donn. Neonatal population pharmacokinetics of phenobarbitol derived from routine clinical data.Dev. Pharmacol. Ther. 8:374–383 (1985).
P. O. Maitre, S. Vozeh, J. Heykants, D. A. Thompson, and D. R. Stanski. Population pharmacokinetics of alfentanil: The average dose-plasma concentration relationship and interindividual variability in patients.Anesthesiology 66:3–12 (1987).
T. H. Grasela, E. J. Antal, L. Ereshefsky, B. G. Wells, R. L. Evans, and R. B. Smith. An evaluation of population pharmacokinetics in therapeutic trials. Part II. Detection of a drug-drug interaction.Clin. Pharmacol. Ther. 42:443–441 (1987).
S. L. Beal and L. B. Sheiner. Estimating population kinetics.CRC Crit. Rev. Biomed. Eng. 8:195–222 (1982).
L. B. Sheiner and S. L. Beal. Evaluation of methods for estimating population pharmacokinetic parameters. I. Michaelis-Menten model: Routine clinical pharmacokinetic data.J. Pharmacokin. Biopharm. 8:553–571 (1980).
L. B. Sheiner and S. L. Beal. Evaluation of methods for estimating population pharmacokinetic parameters. II. Biexponential model and experimental pharmacokinetic data.J. Pharmacokin. Biopharm. 9:635–651 (1981).
L. B. Sheiner and S. L. Beal. Evaluation of methods for estimating population pharmacokinetic parameters. III. Monoexponential model: routine clinical pharmacokinetic data.J. Pharmacokin. Biopharm. 11:303–319 (1983).
E. L. Lehmann,Nonparametrics, Statistical Methods Based on Ranks, Holden-Day, San Francisco, 1975, pp. 81–91.
SAS User's Guide: Basics, Version 5 Edition, SAS institute, Inc., Cary NO, 1985, p. 1187.
E. S. Moore, R. G. Faix, R. C. Banagale, and T. H. Grasela. The population pharmacokinetics of theophylline in neonates and young infants.J. Pharmacokin. Biopharm. 17:47–66 (1989).
E. J. Antal, T. H., Grasela and R. B. Smith. An evaluation of population pharmacokinetics in therapeutic trials. Part III. Prospective data collection versus retrospective data assembly.Clin. Pharmac. Ther. 46:552–559 (1989).
R. B. Smith, P. D. Kroboth, J. T. Vanderlugt, J. P. Phillips, and R. P. Juhl. Pharmacokinetics and pharmacodynamics of alprazolam after oral and iv administration.Psychopharmacology 84:452–456 (1984).
D. Ciraulo, J. G. Barnhill, H. G. Boxenbaum, D. J. Greenblatt, and R. B. Smith. Pharmacokinetic and clinical effects of alprazolam following single and multiple oral doses. in patients with panic disorder.J. Clin. Pharmacol. 26:292–298 (1986).
S. L. Beal, L. B. Sheiner.NONMEM User's Guides, Pan VI, NONMEM Project Group, University of California, San Francisco, 1989, pp. 8–25.
L. B. Sheiner and S. L. Beal. A note on confidence intervals with extended least square parameter estimates.J. Pharmacokin. Biopharm. 15:93–98 (1987).
C. R. Rao.Linear Statistical Inference and Its Applications, Wiley, New York, 1965, pp.347–352.
Author information
Authors and Affiliations
Additional information
Partial support received from Upjohn Co., NIH-BRSG SO RR 07066, and the Burroughs Wellcome Foundation.
Rights and permissions
About this article
Cite this article
White, D.B., Walawander, C.A., Tung, Y. et al. An evaluation of point and interval estimates in population pharmacokinetics using Nonmem analysis. Journal of Pharmacokinetics and Biopharmaceutics 19, 87–112 (1991). https://doi.org/10.1007/BF01062194
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01062194