Summary
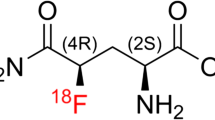
We observed the tissue delivery of a novel water-soluble nitrosourea, 1-(2-chloroethyl)-3-(methyl-α-D-glucopyranos-6-yl)-1-nitrosourea (MCNU) in rats bearing experimental brain tumors by conducting autoradiography on all. Prior to this study, the development of a streaming phenomenon was ascertained (and thus finding the optimum velocity for intra-arterial infusion) by14C-iodoantipyrine (IAP) autoradiography. Furthermore, a single pass extraction value of MCNU was measured. At an arterial infusion rate of 0.2 ml/min., the streaming phenomenon was recognized but the tracer was fairly evenly distributed at a rate of 1.0 ml/min. On the other hand, the single pass extraction value for MCNU was 0.18 ± 0.036 (mean ±S.D., n=3, under pentobarbital anesthesia). It was suggested that MCNU is very unlikely to be transported into the normal rat brain. We conducted14C-MCNU autoradiography to observe tissue distribution of MCNU following its intra-arterial and intravenous infusions in a brain tumor model using rats. The normal side (the side where no infusions were given) and the cerebral cortex at the side affected by the tumor (the side where the infusion was given) showed hardly any uptake of14C-MCNU in both the intra-arterial and intravenous infusion groups. The tumorous section was divided into the periphery and the center to measure tissue concentration of the tracer in each section. Compared against the cortical section, the periphery and the center showed significant increases in the concentration (approximately 11 to 15 times and 3 to 7 times, respectively, the figure for the cortical region) for both the intra-arterial and intravenous groups. When compared against the intravenous infusion group, the arterial infusion group showed a significantly high rate of accumulation (1.3 to 3.9 times).
Similar content being viewed by others
References
Levin VA, Kabra P: Effectiveness of the nitrosoureas as a function of their lipid solubility in the chemotherapy of experimental rat brain tumors. Cancer Chemother Rep 58: 787–792, 1974
Levin VA, Shapiro WR, Clancy TP, Oliverio VT: The uptake distribution, and antitumor activity of 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea in the murine glioma. Cancer Res 30: 2451–2455, 1970
Rosenblum ML, Knebel KD, Vasquez DA, Wilson CB: Brain-tumor therapy. Quantitative analysis using a model system. J Neurosurg 46: 145–154, 1977
Tator CH, Day A, Ng R, Liberman L: Chemotherapy of an experimental glioma with nitrosoureas. Cancer Res 37: 476–481, 1977
Rosenblum ML, Reynolds AF, Smith KA, Rumack BH, Walker MD: Chloroethyl-cyclohexyl-nitrosourea (CCNU) in the treatment of malignant brain tumors. J Neurosurg 39: 306–314, 1973
Wilson CB, Gutin P, Boldrey EB, Crafts D, Levin VA, Enot KJ: Single-agent chemotherapy of brain tumors. Arch Neurol 33: 739–744, 1976
Suzuki M, Hori K, Abe I, Sato H: A new approach to cancer chemo-therapy: selective enhancement of tumor blood flow with angiotensin II. J Natl Cancer Inst 67: 663–669, 1981
Muraoka K: [ACNU delivery to malignant brain tumor tissue and serum: route of administration and combined use of phénobarbital.] Brain and Nerve (Tokyo) 35:1199–1206, 1983 (Jpn)
Neuwelt EA, Frenkel EP, Diehl J, Vu LH, Rapoport S, Hill S: Reversible osmotic blood-brain barrier disruption in humans: Implications for the chemotherapy of malignant brain tumors. Neurosurgery 7: 44–52, 1980
Miyagami M, Kagawa Y, Tsubokawa T: ACNU delivery to malignant glioma tissue by osmotic blood brain barrier modification with intracarotid infusion of hyperosmoral mannitol. Neurological Surgery (Tokyo) 13: 955–963, 1985 (Jpn)
Levin VA, Kabra PM, Freeman-Dove MA: Pharmacokinetics of intra-carotid artery14C-BCNU in the squirrel monkey. J Neurosurg 48: 587–593, 1978
Mori T, Mineura K, Katakura R: [A consideration on pharmacokinetics of a new water-soluble antitumor nitrosourea, ACNU, in patients with malignant brain tumor.] Brain and Nerve (Tokyo) 31: 601–606, 1979 (Jpn)
Harada K, Kiya K, Uozumi T: Pharmacokinetics of a new water-soluble nitrosourea derivative (ACNU) in human gliomas. Surg Neurol 15: 410–414, 1981
Yamashita J, Handa H, Tokuriki Y, Ha YS, Otsuka S, Suda K, Taki W: Intra-arterial ACNU therapy for malignant brain tumors. Experimental studies and preliminary clinical results. J Neurosurg 59: 424–430, 1983
Greenberg HS, Ensminger WD, Chandler WF, Layton PB, Junck L, Knake J, Vine AK: Intra-arterial BCNU chemotherapy for treatment of malignant gliomas of the central nervous system. J Neurosurg 61: 423–429, 1984
Blacklock JB, Wright DC, Dedrick RL, Blasberg RG, Lutz RJ, Doppman JL, Oldfield EH: Drug streaming during intra-arterial chemotherapy. J Neurosurg 64:284–291, 1986
Lutz RJ, Dedrick RL, Boretos JW, Oldfield EH, Blacklock JB, Doppman JL: Mixing studies during intracarotid artery infusionin vivo andin vitro model. J Neurosurg 64: 277–283, 1986
Saris SC, Wright DC, Oldfield EH, Blasberg RC: Intravascular streaming and variable delivery to brain following carotid artery infusions in the Sprague-Dawley rat. J Cereb Blood Flow Metab 8: 116–120, 1988
Iwasaki M, Okawa H, Ninomiya K, Sekine J, Kimura G: Antitumor activity of 3-(methyl-α-D-glucopyranos-2-yl)-1-(2-chloroethyl)-1-nitrosourea. Gann 66: 347–348, 1975
Sekido S, Ninomiya K, Iwasaki M: Biologic activity of MCNU: A new antitumor agent. Cancer Treat Rep 63: 961–970, 1979
Harada K, Kiya K, Okamoto H, Uozumi T: Anti-tumor effect of a water soluble nitrosourea, MCNU, agaists experimental mouse malignant glioma. J Cancer Chemother 8: 1211–1215, 1981 (Jpn)
Harada K, Kiya K, Okamoto H, Uozumi T: Comparative studies on anti-tumor effects of nitrosoureas, ACNU and MCNU, on experimental brain tumors. Neurol Med Chir (Tokyo) 21: 1017–1023, 1981 (Jpn)
Wakabayasi T, Yoshida J, Inoue I, Kageyama N, Nagata M, Kanzaki M: MCNU effectiveness on brain tumor. Part I: Antitumor activityin vitro on human glioma and neuroblastoma ceU lines. J Cancer Chemother 11: 1098–1106, 1984 (Jpn)
Aizawa S, Sako K, Yonemasu Y: Measurement of cerebral blood flow, cerebral blood volume, and cerebral capillary permeability in glioma-bearing rats. Neurol Med Chir (Tokyo) 30: 113–118, 1990
Kaneko S, Abe H, Aida T, Tsuru M, Kodama T, Kobayashi H: Experimental study of immunochemotherapy of brain tumors. Experimental brain tumor model and immunoche-motherapy by a combination of PSK and ACNU. Neurol Med Chir (Tokyo) 20: 997–1005, 1980 (Jpn)
Pellegrino LJ, Cushman AJ: A stereotaxic atlas of the rat brain. Meredith Publishing C., New York, USA, 1967
Crone C: The permeability of brain capillaries to nonelectrolytes. Acta Physiol Scand 64: 407–417, 1965
Shapiro WR, Young DF: Treatment of malignant glioma. A controlled study of chemotherapy and irradiation. Arch Neurol 33: 494–500, 1976
Edwards MS, Levin VA, Wilson CB: Brain tumor chemotherapy: an evaluation of agents in current use for Phase II and III trials. Cancer Treat Rep 64: 1179–1205, 1980
Arakawa M, Shimizu F, Okada N: Effect of 1-(4-amino-2-methyl-pyrimidine-5-yl)-methyl-3-(2-chloroethyl)-3-nitrosourea hydrochloride on leukemia L-1210. Gann 65: 191, 1974
Kapp J, Vance R, Parker JL, Smith RL: Limitation of high dose intra-arterial 1,3-bis(2-chloroethyl)-1-nitrosourea (BCNU) chemotherapy for malignant gliomas. Neurosurgery 10: 715–719, 1982
Phillips TW, Chandler WF, Kindt GW, Ensminger WD, Greenberg HS, Seeger JF, Doan KM, Gyves JW: New implantable continuous administration and bolus dose intracarotid drug delivery system for the treatment of malignant gliomas. Neurosurgery 11: 213–218, 1982
Shingleton BJ, Beinfang DC, Albert DM, Ensminger WD, Chandler WF, Greenberg HS: Ocular toxicity associated with high-dose carmustine. Arch Ophthalmol 100:1766–1772, 1982
Omojora MF, Fox AJ, Auer RN: Hemorrhagic encephalitis produced by selective non-occlusive intracarotid BCNU injection in dogs. J Neurosurg 57: 791–796, 1982
Sako K, Yonemasu Y, Okazaki S, Hodozuka A, Fujita T, Daita G, Takei H: Diffuse low density in the white matter on CT scan after intra-carotid ACNU chemotherapy. Neurol Med Chir (Tokyo) in press
Kanamaru R, Sato H, Asamura M, Kakuta H, Wakui A: The mechanism of action of 1-(2-chloroethyl)-3-(methyl-α-D-glucopyranos-6-yl)-1-nitrosourea (MCNU) in cultured HeLa S3 cells. J Cancer Chemother 8: 877–884, 1981 (Jpn)
Yamada K, Ushio F, Hayakawa T, Arita N, Huang TY, Nagatani M, Yamada N, Mogami H: Distribution of radiolabelled 1-(4-amino-2-methyl-5-pyrimidinyl)methyl-3-(2-chloroethyl)-3-nitrosourea hydrochloride in rat brain tumor: intraarterial versus intravenous administration. Cancer Res 47: 2123–2128, 1987
Bullard DE, Saris SC, Bigner DD: Carotid artery injections in 40-to 99-g Fischer rats: technical note and evaluation of blood flow by various injection techniques. Neurosurgery 14: 406–411, 1984
Miyagami M, Tazoe M, Tubokawa T: MCNU delivery in to malignant brain tumor and normal brain tissue by intravenous or intraarterial infusion. J Cancer Chemother 17: 275–280, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hodozuka, A., Sako, K., Nakai, H. et al. Delivery of a novel nitrosourea, MCNU, to the brain tissue in glioma-bearing rats. J Neuro-Oncol 15, 79–86 (1993). https://doi.org/10.1007/BF01050267
Issue Date:
DOI: https://doi.org/10.1007/BF01050267