Abstract
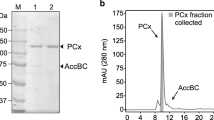
Cells of Clostridium sporosphaeroides which were grown on citrate contained citrate lyase and citrate lyase acetylating enzyme, but no detectable citrate synthase and citrate lyase deacetylase activities. Citrate lyase from C. sporosphaeroides was purified to homogeneity as judged by polyacrylamide gel electrophoresis and high performance liquid chromatography. In contrast to the enzyme from Clostridium sphenoides, the addition of l-glutamate was not necessary for activity and stabilization of the enzyme. The purified enzyme had a specific activity of 34 U/mg protein and was comparable to other citrate lyases with respect to its molecular weight and subunit composition. Electron microscopic investigations showed that similar to the lyase from C. sphenoides and in contrast to all other citrate lyases examined so far, the majority of the enzyme molecules was present in “star” form.
Similar content being viewed by others
References
Antranikian G, Gottschalk G (1982) Copurification of citrate lyase and citrate lyase ligase from Rhodopseudomonas gelatinosa and subsequent separation of the two enzymes. Eur J Biochem 126:43–47
Antranikian G, Giffhorn F, Gottschalk G (1978) Activation and inactivation of citrate lyase ligase from Rhodopseudomonas gelatinosa. FEBS Lett 88:67–70
Antranikian G, Klinner C, Kümmel A, Schwanitz D, Zimmermann T, Mayer F, Gottschalk G (1982) Purification of l-glutamate dependent citrate lyase from Clostridium sphenoides and electron microscopic analysis of citrate lyase isolated from Rhodopseudomonas gelatinosa, Streptococcus diacetilactis and C. sphenoides. Eur J Biochem 126:35–42
Antranikian G, Friese C, Quentmeier A, Hippe H, Gottschalk G (1984) Distribution of the ability for citrate utilization amongst Clostridia. Arch Microbiol 138:179–182
Beuscher N, Mayer F, Gottschalk G (1974) Citrate lyase from Rhodopseudomonas gelatinosa: purification, electron microscopy and subunit structure. Arch Microbiol 100:307–328
Buckel W, Buschmeier V, Eggerer H (1971) Der Wirkungsmechanismus der Citrat-Lyase aus Klebsiella aerogenes. Hoppe-Seyler's Z Physiol Chem 352:1195–1205
Dimroth P, Eggerer H (1975a) Evaluation of the protein compounds of citrate lyase from Klebsiella aerogenes. Eur J Biochem 53:227–235
Dimroth P, Eggerer H (1975b) Isolation of subunits of citrate lyase and characterization of their function in the enzyme complex. Proc Natl Acad Sci (USA) 72:3458–3462
Faloona GR, Srere PA (1969) Escherichia coli citrate synthase. Purification and the effect of potassium and some properties. Biochemistry 8:4497–4503
Giffhorn F, Gottschalk G (1975) Inactivation of citrate lyase from Rhodopseudomonas gelatnosa by a specific deacetylase and inhibition of this inactivation by l(+)glutamate. J Bacteriol 124:1052–1061
Giffhorn F, Beuscher N, Gottschalk G (1972) Regulation of citrate lyase in Rhodopseudomonas gelatinosa. Biochem Biophys Res Commun 49:467–472
Gillespie DG, Gunsalus IC (1953) An adaptive citric desmolase in Streptococcus faecalis. Bacteriol proc p. 80
Gottschalk G, Giffhorn F, Antranikian G (1982) The regulation of citrate lyase by acetylation/deacetylation. Biochem Soc Trans 10:324–326
Kümmel A, Behrens G, Gottschalk G (1975) Citrate lyase from Streptococcus diacetilactis. Association with its acetylating enzyme. Arch Microbiol 102:111–116
Kulla H, Gottschalk G (1977) Energy-dependent inactivation of citrate lyase in Enterobacter aerogenes. J Bacteriol 132:764–770
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Martin RG, Ames BN (1960) A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem 236:1372–1379
Moellering H, Gruber W (1966) Determination of citrate with citrate lyase. Anal Biochem 17:369–376
Peacock AC, Dingman CW (1968) Molecular weight estimation and separation of ribonucleic acid by electrophoresis in agarosepolyacrylamide composite gels. Biochemistry 7:668–674
Siegel LM, Monty KJ (1966) Determination of molecular weights and frictional ratios of proteins in impure systems by use of gel filtration and density gradient centrifugation. Application to crude preparations of sulfite and hydroxylamine reductases. Biochim Biophys Acta 112:346–362
Singh M, Carpenter DE, Srere PA (1974) Nonidentical subunits of citrate lyase from Klebsiella aerogenes. Biochem Biophys Res Commun 59:1211–1218
Valentine RC, Shapiro BM, Stadtman ER (1968) Regulation of glutamine synthetase. XII. Electron microscopy of the enzyme from Escherichia coli. Biochemistry 7:2143–2153
Weber K, Pringle JR, Osborn M (1972) Measurement of molecular weights by electrophoresis on SDS-acrylamide gels. In: Hirs W, Timasheff SN (eds) Methods in enzymology, XXVI. Academic Press, London, pp 3–27
Wheat RW, Ajl SJ (1955) Citritase, the citrate-splitting enzyme from Escherichia coli. Purification and properties. J Biol Chem 217:897–906
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quentmeier, A., Antranikian, G. Characterization of citrate lyase from Clostridium sporosphaeroides . Arch. Microbiol. 141, 85–90 (1985). https://doi.org/10.1007/BF00446745
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00446745