Abstract
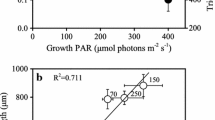
The effect of irradiance on the growth rate, macromolecular composition and photosynthetic carbon metabolism of Amphidinium carteri was studied in batch culture. Growth rate increased linearly with increasing irradiance up to a maximum growth rate of 0.04 h-1 at an irradiance of 80 μEm-2s-1. In contrast to a number of other studies on both prokaryotic and eukaryotic microorganisms, ours showed that cellular content of RNA, DNA, protein and carbohydrate of A. carteri were invariant with growth rate over the range μ=0.04 to 0.007 h-1. The invariant macromolecular composition was correlated with a constant modal cell volume. Chlorophyll and lipid per cell increased with decreasing irradiance. The distribution of [14C]-bicarbonate in the major end products of photosynthesis after incubation with isotope for 14% of a doubling time showed that the percentage carbon in the chloroform (lipids and pigments) fraction increased with decreasing irradiance while that of the trichloroacetic acid soluble (carbohydrate) fractions decreased. The percentage of isotope in the trichloroacetic acid insoluble (protein) fraction and methanol: water fraction (metabolites) remained constant. Because this species, under light-limited growth, differs from other organisms so far studied, more species must be similarly examined before nucleic acid content is used as an index growth rate in the field.
Similar content being viewed by others
References
Blight EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Boehlke KW, Friesen JD (1975) Cellular content of ribonucleic acid and protein in Saccharomyces cerevisiae as a function of exponential growth rate: calculation of the apparent peptide chain elongation rate. J Bacteriol 121:429–433
Brown TE, Richardson FL (1968) The effect of growth environment on the physiology of algae: light intensity. J Phycol 4:38–54
Chan AT (1978) Comparative physiological study of marine diatoms and dinoflagellates in relation to irradiance and cell size. 1. Growth under continuous light. J Phycol 14:396–402
Dorsey TE, McDonald P, Roels OA (1978) Measurements of phytoplankton-protein content with the heated biuret-folin assay. J Phycol 14:167–171
Dortch Q, Roberts TL, Clayton JR Jr, Ahmed SI (1983) RNA/DNA ratios and DNA concentrations as indicators of growth rate and biomass in planktonic marine organisms. Mar Ecol Prog Ser 13:61–71
Franker CK (1970) Some properties of DNA from zooxanthellae harboured by an anenome Anthopleura elegantissima. J Phycol 6:299–305
Galleron C (1976) Synchronization of the marine dinoflagellate Amphidinium carteri in dense cultures. J Phycol 12:69–73
Galleron C, Durrand AM (1978) Characterization of a dinoflagellate (Amphidinium carteri) DNA. Biochimie 60:1235–1242
Galleron C, Durrand AM (1979) Cell cycle and DNA synthesis in a marine dinoflagellate Amphidinium carteri. Protoplasma 100:155–165
Goldman JC, McCarthy JJ, Peavey DG (1979) Growth rate influence on the chemical composition of phytoplankton in oceanic waters. Nature (Lond) 279:210–215
Guillard RRL, Ryther JH (1962) Studies on planktonic diatoms. I. Cyclotella nana Haustedt and Detonula confervacae (Cleve) Gran. Can J Microbiol 8:229–239
Halldall P (1970) The photosynthetic apparatus of microalgae and its adaptation to environmental factors. In: Halldal P (ed) Photobiology of microorganisms. Wiley Interscience, London, pp 17–55
Hitchcock GL (1982) A comparative study of the size-dependent organic composition of marine diatoms and dinoflagellates. J Plankton Res 4:363–377
Holm-Hansen O (1963) Algae: Amounts of DNA and organic carbon in single cells. Science 163:87–88
Holm-Hansen O, Sutcliffe WH Jr, Sharp J (1968) Measurement of deoxyribonucleic acid in the ocean and its ecological significance. Limnol Oceanogr 13:507–514
Kissane JM, Robins E (1958) The fluorimetric measurement of deoxyribonucleic acid in animal tissues with special reference to the central nervous system. J Biol Chem 233:184–188
Koch AR (1980) The inefficiency of ribosomes functioning in Escherichia coli growing at moderate rates. J Gen Microbiol 116:165–171
Kochert G (1978) Carbohydrate determination by the phenolsulfuric acid method. In: Hellebust JA, Craigie JS (eds) Handbook of phycological methods vol 2, Physiological and biochemical methods. Cambridge Univ Press, Cambridge, pp 95–97
Konopka A, Schnur M (1980) Effect of light intensity on macromolecular synthesis in cyanobacteria. Microb Ecol 6: 291–301
Leick V (1967) Growth rate dependency of protein and nucleic acid composition of Tetrahymena pyriformis and the control of synthesis of ribosomal and transfer RNA. C R Trav Lab Carlsberg 46:113–126
Li WKW, Glover HE, Morris I (1980) Physiology of carbon photoassimilation by Oscillatoria thebautii in the Caribbean Sea. Limnol Oceanogr 25:337–456
Loeblich AR III (1976) Dinoflagellate evolution; speculation and evidence. J Protozool 23:13–18
Mann NH, Carr NG (1974) Control of macromolecular composition and cell division in the blue-green alga Anacystis nidulans. J Gen Microbiol 83:399–405
Morris I (1981) Photosynthetic products, physiological state, and phytoplankton growth. In: Platt T (ed) Physiological basis of phytoplankton ecology. Can Bull Fish Aquat Sci 210:83–102
Morris I, Glover HE, Yentsch CS (1974) Products of photosynthesis by marine phytoplankton: the effect of environmental factors on the relative rates of protein synthesis. Mar Biol 27:1–9
Pande SV, Khan RP, Venkitasubramanian TA (1963) Microdetermination of lipids and serum total fatty acids. Anal Biochem 6:415–423
Parsons TR, Stephens K, Strickland JDH (1961) On the chemical composition of eleven species of marine phytoplankton. J Fish Res Bd Can 18:1001–1016
Plaut BS, Turnock G (1975) Coordination of macromolecular synthesis in the slime mould Physarum polycephalum. Mol Gen Genet 137:211–225
Poyton RO (1973) Effect of growth rate on the macromolecular composition of Prototheca zopfii, a colourless alga which divides by multiple fission. J Bacteriol 113:203–211
Prescott DM (1976) Reproduction of eukaryotic cells. Academic Press, New York
Reeves ME, Jones LW (1981) Effect of light intensity on ultrastructure and lipid content of photosynthetic membranes of Fremyella diplosiphon (Cyanophyceae). J Phycol (Suppl) 17:13
Richardson K, Beardall J, Raven JA (1983) Adaptation of unicellular algae to irradiance: an analysis of strategies. New Phytol 93:157–191
Rizzo PJ, Nooden LD (1973) Isolation and chemical composition of dinoflagellate nuclei. J Protozool 20:666–672
Sakshaug E, Myklestad S, Krogh T, Westin G (1973) Production of protein and carbohydrate in the dinoflagellate Amphidinium carteri. Some preliminary results. Norw J Bot 20:211–218
Schaechter M, Maaloe O, Kjeldgaard NO (1958) Dependency on medium and temperature of cell size and chemical composition during balanced growth of Salmonella typhimurium. J Gen Microbiol 19:592–606
Schneider WC (1957) Determination of nucleic acids in tissues by pentose analysis. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 3. Academic Press, London, pp 680–684
Skjold AC, Juarez H, Hedgcoth C (1973) Relationship among deoxyribonucleic acids in Escherichia coli 15 T-at various growth rates. J Bacteriol 115:177–187
Taggart J (1967) Heptulose phosphate synthesis and the transaldolase reaction in plant tissues. Ph. D. Thesis, University of Liverpool
Vernon LP (1960) Spectrophotometric determination of chlorophylls and phenophytins in plant extracts. Anal Chem 32:1144–1150
Waldron O, Lacroute F (1975) Effect of growth rate on the amount of ribosomal and transfer ribonucleic acid in yeast. J Bacteriol 122:855–865
Werner-Schlenzka H, Werner E, Kroger H (1978) Dinoflagellate ribosomal RNA, an early stage in the evolution of eukaryotic RNA. Comp Biochem Physiol 61 B:587–591
Author information
Authors and Affiliations
Additional information
This paper is presented with our best wishes on the occasion of Professor G. Drews 60th birthday
Rights and permissions
About this article
Cite this article
Thomas, P.H., Carr, N.G. The invariance of macromolecular composition with altered light limited growth rate of Amphidinium carteri (dinophyceae). Arch. Microbiol. 142, 81–86 (1985). https://doi.org/10.1007/BF00409242
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00409242