Abstract
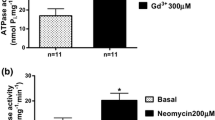
Previous studies in renal cells and hepatocytes have shown that cell swelling leads to a rapid and reversible increase in pH in acidic cellular compartments, including lysosomes. Among the consequences are an inhibition of proteolysis. The present study shows that a similar lysosomal alkalinization occurs upon osmotic swelling of vascular smooth muscle cells, as evidenced by acridine orange and fluorescein isothiocyanate fluorescence. Furthermore, we have studied the mechanism underlying lysosomal alkalinization, which had remained unclear. The lysosomal alkalinization was not abolished by inhibition of vacuolar H+-ATPases (100 nM bafilomycin), Cl− channels [100 μM] 5-nitro-2-(3-phenylpropylamino) benzoic acid (NPPB), carbonic anhydrase (100 μM acetazolamide) or Na+/H+ exchange (10 μM HOE 694). The Ca2+ ionophore A23187 (10 μM) led to a slight increase in lysosomal pH, but removal of extracellular Ca2+ and depletion of cellular Ca2+ stores (100 nM thapsigargin) did not appreciably blunt the swelling-induced lysosomal alkalinization. In the presence of bafilomycin the alkalinizing effect of osmotic cell swelling was not reversible, in contrast to that of NH4Cl. In conclusion, osmotic swelling of vascular smooth muscle cells leads to lysosomal alkalinization, presumably in large part through activation of a hydrogen ion leak.
Similar content being viewed by others
References
Alvarez-Leefmans FJ, Altamirano J, Crowe WE (1995) Measurement of cell water volume changes in single cells using fluorescent probes (abstract). Biophys J 68:A292
Bowman EJ, Siebers A, Altendorf K (1988) Bafilomycins: a class of inhibitors of membrane ATPases from microorganisms, animal cells, and plant cells. Proc Natl Acad Sci USA 85:972–7976
Busch GL, Schreiber R, Dartsch PC, Völkl H, vom Dahl S, Häussinger D, Lang F (1994) Involvement of microtubules in the link between cell volume and pH of acidic cellular compartments in rat and human hepatocytes. Proc Natl Acad Sci USA 91:9165–9169
Cabantchik ZI, Greger R (1992) Chemical probes for anion transporters of mammalian cell membranes. Am J Physiol 262:C803–C827
Geisow MJ (1984) Fluorescein conjugates as indicators of subcellular pH: a critical evaluation. Exp Cell Res 150:29–35
Häussinger D, Lang F (1991) Cell volume in the regulation of hepatic function: a mechanism for metabolic control. Biochim Biophys Acta 1071:331–350
Häussinger D, Lang F (1991) Cell volume — a “second messenger” in the regulation of metabolism by amino acids and hormones. Cell Physiol Biochem 1:121–130
Häussinger D, Lang F (1992) Cell volume and hormone action. Trends Pharmacol 13:371–373
Häussinger D, Roth E, Lang F, Gerok W (1993) Cellular hydration state: an important determinant of protein catabolism in health and disease. Lancet 341:1330–1332
Häussinger D, Lang F, Gerok W (1994) Regulation of cell function by the cellular hydration state. Am J Physiol 267:E343–E355
Lake JR, Van Dyke RW, Scharschmidt BF (1987) Acidic vesicles in cultured rat hepatocytes. Identification and characterization of their relationship to lysosomes and other storage vesicles. Gastroenterology 92:1251–1261
Lang F, Busch GL, Völkl H, Häussinger D (1994) Lysosomal pH — a link between cell volume and metabolism. Biochem Soc Trans 22:502–505
Lang F, Busch GL, Zempel G, Ditlevsen J, Hoch M, Emerich U, Axel D, Fingerle J, Meierkord S, Apfel H, Krippeit-Drews P, Heinle H (1995) Ca2+ entry and vasoconstriction during osmotic swelling of vascular smooth muscle cells. Pflügers Arch 431:253–258
Maren TH (1967) Carbonic anhydrase: chemistry, physiology, and inhibition. Physiol Rev 47:595–781
McCarty NA, O'Neil RG (1992) Calcium signalling in cell volume regulation. Physiol Rev 72:1037–1061
Mortimore GE, Pösö AR (1987) Intracellular protein catabolism and its control during nutrient deprivation and supply. Annu Rev Nutr 7:539–564
Muallem S, Zhang B-X, Loessberg PA, Star R (1992) Simultaneous recording of cell volume changes and intracellular pH or Ca2+ concentration in single osteosarcoma cells UMR-106-01. J Biol Chem 267:17658–17664
Ohkuma S, Poole B (1978) Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci USA 75:3327–3331
Okada Y, Hazama A, Hashimoto A, Maruyama Y, Kubo M (1992) Exocytosis upon osmotic swelling in human epithelial cells. Biochim Biophys Acta 1107:201–205
Ritter M, Paulmichl M, Lang F (1991) Further characterization of volume regulatory decrease in cultured renal epitheloid (MDCK) cells. Pflügers Arch 418:35–39
Ross PE, Garber SS, Calahan MD (1994) Membrane chloride conductance and capacitance in Jurkat T lymphocytes during osmotic swelling. Biophys J 66:169–178
Sabolic I, Burckhardt G (1983) Proton pathways in rat renal brush-border and basolateral membranes. Biochim Biophys Acta 734:210–220
Schreiber R, Stoll B, Lang F, Häussinger D (1994) Effects of anioo-osmolarity and hydroperoxides on intracellular pH in isolated rat hepatocytes as assessed by (2′,7′)-bis(carboxyethyl)-5(6)-carboxyfluorescein and fluorescein isothiocyanate-dextran fluorescence. Biochem J 303:113–120
Tauc M, Le Maout S, Poujeol P (1990) Fluorescent videomicroscopy study of regulatory volume decrease in primary culture of rabbit proximal convoluted tubule. Biochim Biophys Acta 1052:278–284
Thastrup O, Cullen PJ, Drobak BK, Hanley MR, Dawson AP (1990) Thapsigargin, a tumor promoter, discharges intracellular Ca2+ stores by specific inhibition of the endoplasmic reticulum Ca2+-ATPase. Proc Natl Acad Sci USA 87:2466–2470
Thomas JA, Buchsbaum RN, Zimniak A, Racker E (1979) Intracellular pH measurements in Ehrlich ascites tumor cells utilizing spectroscopic probes generatedin situ. Biochemistry 18:2210–2218
Van Dyke RW, Belcher J (1994) Acidification of three types of liver endocytotic vesicles: similarities and differences. Am J Physiol 266:C81–C94
Van Dyke RW, Hornick CA, Belcher J, Scharschmidt BF, Havel RJ (1985) Identification and characterization of ATP-dependent proton transport by rat liver multivesicular bodies. J Biol Chem 260:11021–11026
Völkl H, Friedrich F, Häussinger D, Lang F (1993) Effect of cell volume on acridine orange fluorescence in hepatocytes. Biochem J 295:11–14
Völkl H, Rehwald W, Waitz W, Häussinger D, Lang F (1993) Acridine orange fluorescence in renal proximal tubules: effects of NH3/NH +4 ; and cell volume. Cell Physiol Biochem 3:28–33
Völkl H, Busch GL, Häussinger D, Lang F (1994) Alkalinization of acidic cellular compartments following cell swelling. FEBS Lett 338:27–30
Walker LN, Bowen-Pope DF, Ross R, Reidy MA (1986) Production of platelet-derived growth factor-like molecules by cultured arterial smooth muscle cells accompanies proliferation after arterial injury. Proc Natl Acad Sci USA 83:7311–7315
Warnock D, Reenstra WW, Yee VJ (1982) Na+/H+ antiporter of brushborder vesicles: studies with acridine orange uptake. Am J Physiol 242:F733–F739
Weiss H, Lang F (1992) Ion channels activated by swelling of Madin Darby Canine Kidney (MDCK) cells. J Membr Biol 126:109–114
Wöll E, Ritter M, Offner F, Lang H-J, Schölkens B, Häussinger D, Lang F (1993) Effects of HOE 694 — a novel inhibitor of Na+/H+ exchange - on NIH 3T3 fibroblasts expressing the RAS oncogene. Eur J Pharmacol 246:269–273
Yoshimori T, Yamamoto A, Moriyama Y, Futai M, Tashiro Y (1991) Bafilomycin A1, a specific inhibitor of vacuolar-type H+-ATPase, inhibits acidification and protein degradation in lysosomes of cultured cells. J Biol Chem 266:17707–17712
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Busch, G.L., Lang, HJ. & Lang, F. Studies on the mechanism of swelling-induced lysosomal alkalinization in vascular smooth muscle cells. Pflugers Arch. 431, 690–696 (1996). https://doi.org/10.1007/BF02253831
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02253831