Abstract
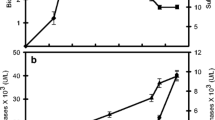
The growth of the microorganism and the production of the pectinolytic enzyme complex in a stirred 30-l biofermentor using the Aspergillus niger Rehbrücke strain were studied. The time courses of fermentation parameters (formation of biomass, consumption of carbon and inorganic nitrogen source, formation of pectinolytic enzymes) were measured. The formation of biomass showed a distinct lag phase, followed by a log phase with exponential growth and finally a stationary period when cell lysis was beginning. The uptake of the carbon source and inorganic nitrogen source by the A. niger cells corresponded to the time course of growth. The formation of pectinolytic enzymes took place in two steps. The first one was growth-bounded and finished with the end of the log phase of biomass growth. The second step of pectinolytic enzyme formation took place after the end of the catabolite repression of the carbon source and was not growth-bounded. On the basis of the experimental data a mathematical model of the fermentation process was developed. Comparison of the kinetics of the measured fermentation curves and the solution curves of the model showed qualitatively good agreement.
Similar content being viewed by others
References
Aguilar G, Huitron C (1986) Application of fed-batch cultures in the production of extracellular pectinases by Aspergillus sp. Enzyme Microb Technol 8:541–545
Aguilar G, Huitron C (1987) Stimulation of the production of extracellular pectinolytic activities of Aspergillus sp. by galacturoni acid and glucose addition. Enzyme Microb Technol 9:690–696
Blieva RK, Rodionova NA (1987) Fractionation and purification of pectin-degrading enzymes by immobilized cells of Aspergillus awamore. Prikl Biokhim Mikrobiol 23:561–567
Kester HCM, Visser J (1990) Purification and characterization of polygalacturonases produced by the hyphal fungus Aspergillus niger. Biotechnol Appl Biochem 12:150–160
Lange R, Friebe R, Linow F, Schaffner B (1979) Zur Methodenkombination Kjeldahl-Aufschluß/Berthelot-Reaktion bei der Stickstoffbestimmung in biologischen Materialien 2. Mitt Nahrung 23:431–439
Larios G, Garcia JM, Huitron C (1989) Endo-polygalacturonase production from untreated lemon peel by Aspergillus sp. CH-Y-1043. Biotechnol Lett 11:729–734
Maldonado MC, Strasser de Saad AM, Callieri D (1989) Catabolite repression of the synthesis of inducible polygalacturonase and pectinesterase by Aspergillus niger sp. Curr Microbiol 18:303–306
Maldonado MC, Strasser de Saad AM, Callieri D (1994) Purification and characterization of pectinesterase produced by a strain of Aspergillus niger. Curr Microbiol28:193–196
Monod J (1942) La croissance des cultures bacteriennes. Hermann, Paris
Rombouts FM, Pillnik W (1980) Pectic enzymes. In: Rose AH (ed) Microbial enzymes and bioconversions. Academic Press, London, pp 227–282
Shinmyo A, Davis L, Nomoto T, Tahara T, Enatsu T (1978) Catabolite repression of hydrolases in Aspergillus niger. Eur J Appl Microbiol Biotechnol 5:59–68
Tahara T, Doi S, Shinmyo A, Terni G (1972) Translational repression in the preferential synthesis of some mould enzymes. 1. J Ferment Technol 50:655–661
Yurkerich VV (1985) Feed back control of synthesis of secreted enzymes by their level in the medium. In: Kulaev IS, Dawes EA, Tempest DW (eds) Environmental regulation of microbial metabolism. Proceedings of the Federation of European Microbiological Societies Symposium. Pushchino, USSR, 1983. Academic Press, New York London, pp 317–321
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, O., Angermann, H., Frommhold-Treu, I. et al. Experimental and theoretical investigations of submerged fermentation and synthesis of pectinolytic enzymes by Aspergillus niger . Appl Microbiol Biotechnol 43, 424–430 (1995). https://doi.org/10.1007/BF00218444
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00218444