Abstract
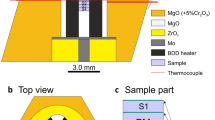
In situ X-ray diffraction study on KAlSi3O8 has been performed using the cubic type high pressure apparatus, MAX90, combined with synchrotron radiation. We determined the phase relations of sanidine, the wadeite-type K2Si4O9+kyanite (Al2SiO5)+coesite (SiO2) assemblage, and hollandite-type KAlSi3O8, including melting temperatures of potassic phases, up to 11 GPa. Our data on subsolidus phase boundaries are close to the recent data of Yagi and Akaogi (1991). Melting relations of sanidine are consistent with the low pressure data of Lindsley (1966). The breakdown of sanidine into three phases reduces melting temperature, and wadeite-type K2Si4O9 melts first around 1500° C in three phase coexisting region. Melting point of hollandite-type KAlSi3O8 is between 1700° C and 1800° C at 11 GPa. If these potassic phases host potassium in the earth's mantle, the true mantle solidus temperature will be much lower than the reported dry solidus temperature of peridotite.
Similar content being viewed by others
References
Decker DL (1971) High-pressure equation of state for NaCl, KCl, and CsCl. J Appl Phys 42:3239–3244
Harlow GE, Veblen DR (1991) Potassium in clinopyroxene inclusions from diamonds. Nature 251:652–655
Kanzaki M, Kurita K, Fujii T, Kato T, Shimomura O, Akimoto S (1987) A new technique to measure the viscosity and density of silicate melts at high pressure. In: High Pressure Research in Mineral Physics, edited by M. Manghnani and Y. Syono, TERRAPUB, Tokyo/AGU, Washington, D.C., pp 195–200
Kinomura N, Kume S, Koizumi M (1975) Stability of K2Si4O9 with wadeite type structure. In: Proc. 4th Inter. Conf. on High Pressure, pp 211–214
Lindsley DH (1966) Melting relations of KAlSi3O8: effect of pressures up to 40 kilobars. Am Mineral 51:1793–1799
Liu L-G (1987) High-pressure phase transitions of potassium aluminosilicates with an emphasis on leucite. Contrib Mineral Petrol 95:1–3
Ringwood AE, Reid AF, Wadsley AD (1967) High-pressure KAlSi3O8, an aluminosilicate with sixfold coordination. Acta Cryst 23:1093–1095
Shimomura O, Utsumi W, Taniguchi T, Kikegawa T, Nagashima T (1992) A new high pressure and high temperature apparatus with sintered diamond anvils for synchrotron radiation use. In: High Pressure Research: Application to Earth and Planetary Sciences, edited by Y Syono and M Manghnani, TERRA-PUB, Tokyo/AGU, Washington, D.C., pp 3–11
Somerville M, Ahrens TJ (1980) Shock compression of KFeS2 and question of potassium in the core. J Geophys Res 85:7016–7024
Sykes D, Poe BT, McMillan PF, Luth RW, Sato RK (1993) A spectroscopic investigation of anhydrous KAlSi3O8 and NaAlSi3O8 glasses quenched from high pressure. Geochim Cosmochim Acta 57:1753–1759
Takahashi E, Kushiro I (1983) Melting of a dry peridotite at high pressures and basalt magma genesis. Am Mineral 68:859–879
Takahashi E (1986) Melting of a dry peridotite KLB-1 up to 14 GPa: implications on the origin of peridotitic upper mantle. J Geophys Res 91:9367–9382
Wendlandt RF, Eggler DH (1980) The origins of potassic magmas: 1. melting relations in the systems KAlSiO4-Mg2SiO4-SiO2 and KAlSiO4-MgO-SiO2-CO2 to 30 kilobars. Am J Sci 280:385–420
Yagi A, Akaogi M (1991) Phase transition of KAlSi3O8. In: Program and abstracts of papers of the 32th high pressure conf. Japan 1991, pp 70–71
Yagi T, Akimoto S (1976) Direct determination of coesite-stishovite transition by in situ X-ray measurements. Tectonophysics 35:259–270
Yamada H, Matsui Y, Ito E (1984) Crystal-chemical characterization of KAlSi3O8 with the hollandite structure. Mineral J 12:29–34
Yamaoka S, Shimomura O, Yagi T, Akimoto S (1985) Direct observation of the conversion reaction from graphite to diamond. In: Sol State Phys under Pressure: Recent Advance with Anvil Devices, edited by S. Minomura, KTK Scientific Publishers/Tokyo, pp 369–374
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Urakawa, S., Kondo, T., Igawa, N. et al. Synchrotron radiation study on the high-pressure and high-temperature phase relations of KAlSi3O8 . Phys Chem Minerals 21, 387–391 (1994). https://doi.org/10.1007/BF00203296
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00203296