Abstract
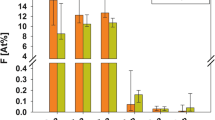
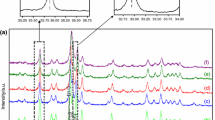
It is stated that all the synthetic carbonate-hydroxyapatites, produced with the reaction of H3PO4 and Ca(OH)2 solutions, are B type carbonate-apatites. The carbonate content of these is completely eliminated up to 900° C. In dental enamels taken from the healthy teeth of females, the carbonate ions occupy different positions (A–B type carbonate-apatite). Those which are parallel to axis c are decomposed only above 900° C. It was found that the natural ageing of dental enamels is in correlation with the different CO 2−3 /H2O ratio measured in these samples. Similar differences were observed between enamels taken from different parts of the tooth and the dentin. The change of the apatite composition after fluoride painting has been established by infrared (IR), derivatographic and X-ray methods.
Similar content being viewed by others
References
Bett, J.A.S., Christner, L.G., Hall, W.K.: Studies of the hydrogen held by solids. XII. Hydroxyapatite catalysits. J. Am. Chem. Soc. 89, 5535–41 (1967)
González-Diaz, P.F., Hidalgo, A.: Infrared spectra of calcium apatites. Spectrochim. Acta 32A, 631–35 (1976)
Matthews, A., Nathan, Y.: The decarbonation of carbonate-fluroapatite (francolite). Am. Mineral. 62, 565–73 (1977)
McConnell, D.: Apatite: its crystal chemistry, mineralogy, utilization and geologic and biologic occurrences. Wien, New York: Springer 1973
McConnell, D., Foreman, D.W.: Model for carbonate apatite. Inorg. Chem. 17, 2039–40 (1978)
Roux, P., Bonel, G.: Étude par diffraction des rayons X et par spectrométrie d'absorption infrarouge des apatites carbonatées de type A phospho-calcique et arsénio-calcique haute pression. Bull. Minéral. 101, 448–52 (1978)
Santos, M., González-Diaz, P.F.: A model for B carbonate apatite. Inorg. Chem. 16, 2131–34 (1977)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jónás, K., Vassányi, I. & Ungvári, I. The study of synthetic carbonate-hydroxyapatites and dental enamels by IR and derivatographic methods. Phys Chem Minerals 6, 55–60 (1980). https://doi.org/10.1007/BF00308394
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00308394