Abstract
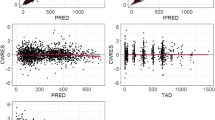
The pharmacokinetics of 222 infusions of high-dose methotrexate (MTX) with leucovorin rescue were studied in 22 adults with osteosarcoma. To reduce the variability of plasma concentration, we individualized dose regimens using a Bayesian method to reach a concentration of 10−3 M MTX at the end of an 8-h infusion. The mean concentration observed at the end of the infusion was 1016±143 μmol/l. The mean dose delivered was 13.2±2 g/m2. The clearance was 49.1±11.7 ml min−1 m−2. The decay of the plasma concentration of MTX after completion of the infusion followed a two-compartment model with at 1/2α of 2.66±0.82 h and at 1/2β of 15.69±8.63 h. The volume of distribution was 0.32±0.08 l/kg. As compared with previously published data, the interindividual and intraindividual variations in the concentration at the end of the infusion were reduced, with values of 14% and 5.9%–21%, respectively, being obtained. Severe toxicities were avoided, and there were only 3 hematologic and 8 digestive grade 3 side effects and no grade 4 complication. Thet 1/2α and the MTX plasma concentrations at 23 and 47 h were correlated with renal toxicity (P<0.001). However, no correlation was found between the pharmacokinetic parameters and other signs of toxicity. There was no significant difference in pharmacokinetics between the toxic and nontoxic groups. In the same manner, the parameters of the group of patients sensitive to MTX were not statistically significantly different from those of the group of nonsensitive patients.
Similar content being viewed by others
References
Bard Y (1974) Non linear parameter estimation. Academic Press, New York
Bisset D, Kaye S, Kerr J (1991) Can primary osteosarcoma act as a third space after high dose methotrexate? Eur J Cancer 27: 1060
Borsi J, Moe PJ (1987) Systemic clearance of methotrexate in the prognosis of acute lymphoblastic leukemia in children. Cancer 60: 3020
Borsi J, Schullet D, Moe PJ (1988) Methotrexate administered by 6 h and 24 h infusion: pharmacokinetic comparison. Cancer Chemother Pharmacol 22: 33
Breithaupt H, Kuenzlen E (1982) Pharmacokinetics of methotrexate and 7-hydroxymethotrexate following infusions of high dose methotrexate. Cancer Treat Rep 66: 1733
Bruno R, Iliadis A, Favre R, Lena N, Imbert AM, Cano JP (1985) Dosage predictions in high-dose methotrexate infusions. 2: Bayesian estimation of methotrexate clearance. Cancer Drug Deliv 2: 277
Calvert AH, Bondy PK, Harrap KR (1977) Some observations on the human pharmacology of methotrexate. Cancer Treat Rep 61: 1647
Campbell MA, Perrier DG, Dorr RT, Alberts DS, Finley PR (1985) Methotrexate: bioavailability and pharmacokinetics. Cancer Treat Rep 69: 833
Chabner BH, Young RC (1973) Threshold methotrexate concentration for in vivo inhibition of DNA synthesis in normal and tumor target tissues. J Clin Invest 52: 1804
Chatelut E, Roche H, Pusquellec Y, Peyrille F, De Biasi J, Pujol A, Canal P, Houin G (1991) Pharmacokinetic modeling of plasma and cerebrospinal fluid methotrexate after high dose intravenous infusion in children. J Pharm Sci 80: 730
Delepine N, Delepine G, Desbois JC (1991) A monocentric therapy study: an approach to optimize the results of the treatment of osteosarcoma by protocols based upon HDMTX associated with systematic conservative surgery in osteosarcoma in adolescents and young adults. G. Bennet Humphrey, Boston
Evans WE, Pratt CB, Taylor RH, Barker LF, Crom WR (1979) Pharmacokinetic monitoring of methotrexate. Cancer Chemother Pharmacol 3: 161
Iliadis A (1985) APIS: a computer program for clinical pharmacokinetics. J Clin Pharmacol 4: 573
Iliadis A, Bruno R, Cano JP (1986) Steady state dosage regimen calculation in linear pharmacokinetics. Int J Biomed Comput 18: 167
Iliadis A, Bruno R, Cano JP (1988) Dynamical dosage regimen calculations in linear pharmacokinetics. Comput Biomed Res 21: 203
Imbert AM, Pignon T, Lena N (1983) Methotrexate assay by enzymatic inhibition: comparison between centrifugal analysis (cobas bio) and competitive protein binding assay. Clin Chem 29: 1317
Jaffe N (1972) Recent advance in the chemotherapy of metastatic osteogenic sarcoma. Cancer 30: 1627
Luyck M, Carin JL, Brunet C, Gosselin P, Demaille MC (1985) Clinical pharmacokinetics of 6-hour infusions of high dose methotrexate. Preliminary trial of monitoring high infusion doses. Eur J Clin Pharmacol 28: 457
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47: 207
Monjanel S, Rigault JP, Cano JP, Carcassonne Y, Favre R (1979) High dose methotrexate: preliminary evaluation of pharmacokinetic approach. Cancer Chemother Pharmacol 3: 189
Niremberg A, Mosende C, Mehta BM, Gisolfi AL, Rosen G (1977) High dose methotrexate with citrovorum factor rescue: predictive value of serum methotrexate concentration and corrective measures to avert toxicity. Cancer Treat Rep 61: 779
Paxton JW (1982) The protein binding and elimination of methotrexate after intravenous infusions in cancer patients. Clin Exp Pharmacol Physiol 9: 225
Pearson AD, Amineddine HA, Yule M, Mills S, Long DR, Craft AW, Chesbells JM (1991) The influence of serum methotrexate concentrations and drug dosage on outcome in childhood acute lymphoblastic leukaemia. Br J Cancer 64: 169
Pittman SW, Parker LM, Tatersall MH (1975) Clinical trial of high dose methotrexate (NRC-740) with citrovorum factor (NSC-3590), toxicologic and therapeutic observation. Cancer Chemother Rep 61: 43
Raude E, Oellerich M, Weinel P, Freund M, Schrappe M, Riehm H, Poliwoda H (1988) High dose methotrexate: pharmacokinetics in children and young adults. Int J Clin Pharmacol Ther Toxicol 26: 364
Rosen G, Marcove R, Caparros B, Nirenberg A, Kossloff C, Huvos A (1979) Primary osteogenic sarcoma: the rationale for preoperative chemotherapy and delayed surgery. Cancer 43: 2163
Rosen G, Caparros B, Huvos A, Kosloff C, Niremberg A, Cacavio A, Marcove R, Lane J, Metha B, Urban C (1982) Preoperative chemotherapy for osteogenic osteosarcoma: selection of postoperative adjuvant chemotherapy based on the response of the primary tumor to preoperative chemotherapy. Cancer 49: 1221
Schalhorn A, Sauer H, Wilmans W, Stupp-Poutot G (1983) MTX and MTX polyglutamates in human sarcoma metastases after HD-MTX therapy. Proc 13th Int Congr Chemother 251: 11
Tattersall MHN, Parker LM, Pitman SW, Frei E (1975) Clinical pharmacology of high dose methotrexate (NSC-740). Cancer Treat Rep 6: 25
Wolfrom C, Hepp R, Hartmann R, Breithaupt H, Henze G (1990) Pharmacokinetic study of methotrexate folinic acid and their serum metabolites in children treated with high dose methotrexate and leucovorin rescue. Eur J Clin Pharmacol 39: 377
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pignon, T., Lacarelle, B., Duffaud, F. et al. Pharmacokinetics of high-dose methotrexate in adult osteogenic sarcoma. Cancer Chemother. Pharmacol. 33, 420–424 (1994). https://doi.org/10.1007/BF00686272
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00686272