Summary
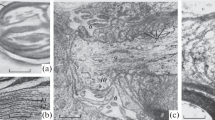
The peripheral nervous system of a mutant of a Japanese quail deficient in neurofilaments (mutant) and of a normal Japanese quail (control) was morphometrically evaluated to characterize the morphological findings, especially those of the myelinated fibers of the mutant. In the proximal peroneal nerves, the frequency of the teased myelinated fibers showing the focal myelin thickening was higher in mutant than in control (P<0.001) without obvious ongoing axonal degeneration and segmental demyelination. The total numbers of the myelinated fibers in the proximal and distal peroneal nerve, and in the tibial nerve branch to gastrocnemius muscle (pars medialis) were similar between control and mutant, although the number of the large myelinated fibers was less (P<0.01) and the number of the small myelinated fibers was greater (P<0.01) in mutant compared with control. The median diameters of neuronal cell bodies of the sacral dorsal root ganglia were similar in control and mutant. The percentages of light, dark and unclassified cells evaluated based on the histologic cytoplasmic features were also similar in control and mutant. Therefore, morphometric alterations were more pronounced in the peripheral myelinated nerve fibers compared with those in the cell bodies of the spinal dorsal root ganglia. We concluded that a smaller number of large myelinated fibers with a greater number of small myelinated fibers and the presence of focal myelin thickening are the main morphologic findings in this mutant, probably due to the arrest of radial growth or maturation of the axons of the myelinated fibers in the absence of ongoing myelinated fiber degeneration.
Similar content being viewed by others
References
Berthold C-H, Skoglund S (1968) Postnatal development of feline paranodal myelin-sheath segments. I. Light microscopy. Acta Soc Med Upsal 73: 113–126
de Waegh SM, Lee VM-Y, Brady ST (1992) Local modulation of neurofilament phosphorylation, axonal caliber, and slow axonal transport by myelinating Schwann cells. Cell 68: 451–463
Dyck PJ (1969) Experimental hypertrophic neuropathy. Pathogenesis of onion-bulb formations produced by repeated tourniquet applications. Arch Neurol 21: 73–95
Dyck PJ (1975) Inherited neuronal degeneration and atrophy affecting peripheral motor, sensory, and autonomic neurons. In: Dyck PJ, Thomas PK, Lambert EH, Bunge R (eds) Peripheral neuropathy, vol 2. WB Saunders, Philadelphia, pp 825–867
Dyck PJ, Stevens JC, Mulder DW, Espinosa RE (1975) Frequency of nerve fiber degeneration of peripheral motor and sensory neurons in amyotrophic lateral sclerosis: morphometry of deep and superficial peroneal nerves. Neurology 25: 781–785
Dyck PJ, Karnes J, Lais AC, Lofgren EP, Stevens JC (1984) Pathologic alterations of the peripheral nervous system of humans. In: Dyck PJ, Thomas PK, Lambert EH, Bunge R (eds) Peripheral neuropathy, vol 2. WB Saunders, Philadelphia, pp 760–870
Dyck PJ, Nukada H, Lais AC, Karnes JL (1984) Permanent axotomy: a model of chronic neuronal degeneration preceded by axonal atrophy, myelin remodeling, and degeneration. In: Dyck PJ, Thomas PK, Lambert EH, Bunge R (eds) Peripheral neuropathy, vol 2. WB Saunders, Philadelphia, pp 666–690
Friede RL, Samorajski T (1970) Axon caliber related to neurofilaments and microtubules in sciatic nerve fibers of rats and mice. Anat Rec 167: 379–388
Griffin JW, Hoffman PN, Clark AW, Carrol PT, Price DL (1978) Slow axonal transport of neurofilament proteins: impairment by β,β′-iminodipropionitrile administration. Science 202: 633–635
Hoffman PN, Griffin JW, Price DL (1984) Control of axonal caliber by neurofilament transport. J Cell Biol 99: 705–714
Hoffman PN, Thompson GW, Griffin JW, Price DL (1985) Changes in neurofilament transport coincide temporally with alterations in the caliber of axons in regenerating motor fibers. J Cell Biol 101: 1332–1340
Hoffman PN, Cleveland DW, Griffin JW, Landes PW, Cowan NJ, Price DL (1987) Neurofilament gene expression: a major determinant of axon caliber. Proc Natl Acad Sci USA 84: 3472–3476
Kawamura Y, Dyck PJ, Shimono M, Okazaki H, Tateishi J, Doi H (1981) Morphometric comparison of the vulnerability of peripheral motor and sensory neurons in amyotrophic lateral sclerosis. J Neuropathol Exp Neurol 40: 667–675
Madrid R, Bradley WG (1975) The pathology of neuropathies with focal thickening of the myelin sheath (tomaculous neuropathy). Studies on the formation of the abnormal myelin sheath. J Neurol Sci 25: 415–448
Mizutani M, Nunoyama T, Yamasaki H, Itakura C (1992) The hypotrophic axonopathy mutant in Japanese quail. J Hered 83: 234–235
Monteiro MJ, Hoffman PN, Gearhart JD, Cleveland DW (1990) Expression of NF-L in both neuronal and nonneuronal cells of transgenic mice: increased neurofilament density in axons without affecting caliber. J Cell Biol 111: 1543–1557
Muma NA, Slunt HH, Hoffman PN (1991) Postnatal increases in neurofilament gene expression correlate with the radial growth of axons. J Neurocytol 20: 844–854
Nukada H, Dyck PJ (1984) Decreased axon caliber and neurofilaments in hereditary motor and sensory neuropathy, type I. Ann Neurol 16: 238–241
Ochoa J, Fowler TJ, Gilliat RW (1972) Anatomical changes in peripheral nerves compressed by a pneumatic tourniquet. J Anat 113: 433–455
Ohnishi A, Dyck PJ (1974) Loss of small peripheral sensory neurons in Fabry disease. Histologic and morphometric evaluation of cutaneous nerves, spinal ganglia, and posterior columns. Arch Neurol 31: 120–127
Ohnishi A, Ogawa M (1986) Preferential loss of large lumbar primary sensory neurons in carcinomatous sensory neuropathy. Ann Neurol 20: 102–104
Ohnishi A, Schilling K, Brimijoin WS, Lambert EH, Fairbanks VF, Dyck PJ (1977) Lead neuropathy. 1. Morphometry, nerve conduction, and choline acetyltransferase transport: new finding of endoneurial edema associated with segmental demyelination. J Neuropathol Exp Neurol 36: 499–518
Ohnishi A, Murai Y, Ikeda M, Fujita T, Furuya H, Kuroiwa Y (1989) Autosomal recessive motor and sensory neuropathy with excessive myelin outfolding. Muscle Nerve 12: 568–575
Raine CS, Wissniewski H, Prineas J (1969) An ultrastructural study of experimental demyelination and remyelination. II. Chronic experimental allergic encephalomyelitis in the peripheral nervous system. Lab Invest 21: 316–327
Tennyson VM (1975) Light and electron microscopy of dorsal root and sympathetic ganglia. In: Dyck PJ, Thomas PK, Lambert EH (eds) Peripheral neuropathy, vol 1. WB Saunders, Philadelphia, pp 74–103
Yamasaki H, Itakura C, Mizutani M (1991) Hereditary hypotrophic axonopathy with neurofilament deficiency in a mutant strain of the Japanese quail. Acta Neuropathol 82: 427–434
Yamasaki H, Bennett GS, Itakura C, Mizutani M (1992) Defective expression of neurofilament protein subunits in hereditary hypotrophic axonopathy of quail. Lab Invest 60: 734–743
Author information
Authors and Affiliations
Additional information
Supported in part by the Research Grant (2A-04) for Nervous and Mental Disorders from the Ministry of Health and Welfare, Japan
Rights and permissions
About this article
Cite this article
Zhao, J.X., Ohnishi, A., Itakura, C. et al. Smaller number of large myelinated fibers and focal myelin thickening in mutant quails deficient in neurofilaments. Acta Neuropathol 86, 242–248 (1993). https://doi.org/10.1007/BF00304138
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304138