Summary
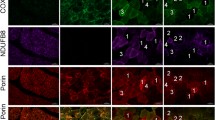
The immunohistochemical reaction of monoclonal as well as polyclonal antibodies against cytochrome c oxidase (COX) subunits with serial sections of normal human skeletal muscle was investigated. The stronger reactivity of polyclonal antibodies to COX subunits II–III and VIIbc with type I as compared to type II fibres, correlated well with the higher histochemical reactivity of NADH dehydrogenase, succinate dehydrogenase and cytochrome c oxidase in type I fibres. In contrast an almost exclusive reaction of a monoclonal antibody against subunit IV with type I fibre and a preponderan reaction of a polyclonal antibody against subunits Vab with type II fibres was obtained. Antibodies against subuntis I, Vb and VIc did not reveal a fibre-type-specific reactivity. The data indicate in human muscle the occurrence of fibre type-specific isozymes of cytochrome c oxidase differing in subunits IV and Va or Vb.
Similar content being viewed by others
References
Büge, U, Kadenbach B (1986) Influence of buffer composition, membrane lipids and proteases on the kinetics of reconstituted cytochrome-c oxidase from bovine liver and heart. Eur. J. Biochem 161:383–390
Brooke MH, Kaiser KK (1970) Three “myosin adenosine triphosphatase” systems: the nature of their pH lability and sulfhydryl dependence. J Histochem Cytochem 18:670–672
Chan SHP, Gai W-Z (1988) Monoclonal antibodies of mammalian cytochrome oxidase and its protein subunits. In: Kim CH, Tedeschi H, Diwan J-J, Salerno JC (eds) Advances in membrane biochemistry and bioenergetics. Plenum, New York, pp 193–202
Dubowitz V, Brooke MH (1973) Muscle biopsy: a modern approach. Saunders, Philadelphia
Greenberg BD, Newbold JE, Sugino A (1983) Intraspecific nucleotide sequence variability surrounding the origin of replication in human mitochondrial DNA. Gene:21:33–49
Hood DA, Zak R, Pette D (1989) Chronic stimulation of rat skeletal muscle induces coordinate increases in mitochondrial and nuclear mRNAs of cytochrome-c-oxidase subunits. Eur J Biochem 179:275–280
Johnson MA, Kadenbach B, Droste M, Old SL, Turnbull DM (1988) Immunocytochemical studies of cytochrome oxidase subunits in skeletal muscle of patients with partial cytochrome oxidase deficiencies. J Neurol Sci 87:75–90
Kadenbach B, Hartmann R, Glanville R, Buse G (1982) Tissue-specific genes for polypeptide VIa of bovine liver and heart cytochrome c oxidase. FEBS Lett 138:236–238
Kadenbach B (1983) Struktur und Evolution des Atmungsferments Cytochrom-c Oxidase (Structure and evolution of the “Atmungsferment” cytochrome-c oxidase). Angew Chem 95:273–281 (Angew Chem Int Edn Engl 22:275–282)
Kadenbach B, Jarausch J, Hartmann R, Merle P (1983) Separation of mammalian cytochrome c oxidase into 13 polypeptides by a sodium dodecyl sulfate-gel electrophoretic procedure. Anal Biochem 129:517–521
Kadenbach B, Stroh A, Ungibauer M, Kuhn-Nentwig L, Büge U, Jarausch J (1986) Isozymes of cytochrome-c oxidase: characterization and isolation from different tissues. Methods Enzymol 126:32–46
Kadenbach B, Kuhn-Nentwig L, Büge U (1987) Evolution of a regulatory enzyme: cytochrome-c oxidase (complex IV). Curr Top Bioenerg 15:113–161
Kadenbach B, Hüther F-J, Büge U, Schlerf A, Johnson MA (1989) Regulatory complexity of cytochrome c oxidase and its defective manifestation in mitochondrial diseases. In: Azzi A, Drahota Z, Papa S (eds) Molecular basis of membrane-associated diseases. Springer, Berlin Heidelberg New York, pp 216–217
Kadenbach B, Stroh A, Becker A, Eckerskorn C, Lottspeich F (1990) Tissue- and species specific expression of cytochrome c oxidase isozymes in vertebrates. Biochim Biophys Acta 1015:368–372
Kuhn-Nentwig L, Kadenbach B (1985) Isolation and properties of cytochrome c oxidase from rat liver and quantification of immunological differences between isozymes from various rat tissues with subunit-specific antisera. Eur J Biochem 149:147–158
Kuhn-Nentwig L, Kadenbach B (1986) Isolation and characterization of human heart cytochrome c oxidase. J Bioenerg Biomembr 18:307–314
Merle P, Jarausch J, Trapp M, Scherka R, Kadenbach B (1981) Immunological and chemical characterization of rat liver cytochrome c oxidase. Biochim Biophys Acta 669:222–230
Monnat RJ, Loeb LA (1985) Nucleotide sequence preservation of human mitochondrial DNA. Proc Natl Acad Sci USA 82:2895–2899
Nakagawa M, Miranda AF, Moggio M, Bonilla E, DiMauro S (1985) Demonstration of tissue-specific cytochrome-c-oxidase (COX) with monoclonal antibodies. Neurology 35 [Suppl 1]:95
Nakagawa M, Miranda AF (1987) A monoclonal antibody against cytochrome c oxidase distinguishes cardiac and skeletal muscle mitochondria. Exp Cell Res 168:44–52
Peters JH, Baumgarten H, Schulze M (1985) Monoklonale Antikörper. Herstellung und Charakterisierung. Springer, Berlin Heidelberg New York Tokyo
Pette D, Urbova G (1985) Invited review: neural control of phenotypic expression in mammalian muscle fibres. Muscle Nerve 8:676–689
Rizzuto R, Nakase H, Zeviani M, DiMauro S, Schon EA (1988) Subunit Va of human and bovine cytochrome c oxidase is highly conserved. Gene 69:245–256
Schlerf A, Droste M, Winter M, Kadenbach B (1988) Characterization of two different genes (cDNA) for cytochrome c oxidase subunit VIa from heart and liver of the rat. EMBO J 7:2387–2391
Seligman AM, Karnousky MJ, Wasserkrug H, Hanker JS (1968) Non-droplet ultrastructural demonstration of cytochrome oxidase activity with a polymerizing osmiophilic reagent, diaminobenzidine (DAB). J Cell Biol 38:1–14
Yanamura W, Zhang Y-Z, Takamiya S, Capaldi RA (1988) Tissue-specific differences between heart and liver cytochrome c oxidase. Biochemistry 27:4909–4914
Zeviani M, Nakagawa M, Herbert J, Lomax MI, Grossman LI, Sherbany AA, Miranda AF, DiMauro S, Schon EA (1987) Isolation of a cDNA clone encoding subunit IV of human cytochrome c oxidase. Gene 55:205–217
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Romero, N., Marsac, C., Fardeau, M. et al. Immunohistochemical demonstration of fibre type-specific isozymes of cytochrome c oxidase in human skeletal muscle. Histochemistry 94, 211–215 (1990). https://doi.org/10.1007/BF02440190
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02440190