Abstract
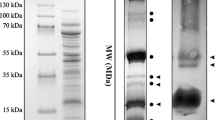
In thylakoid membranes isolated from green plants of parsley, pea, and barley, the light-harvesting chlorophyll a/b protein complex (LHCP, mol. weight: 25,000), is a major constituent. Poly(A)RNA isolated from these species was translated in a wheat germ, cell-free system. The in vitro translation products were treated with antibodies raised against the LHCP. This treatment resulted in the precipitation of a precursor protein (mol. weight: 29,000). Poly(A)RNA was also prepared from a cell culture ofPetroselinum that does not develop chloroplasts upon illumination. This poly(A)RNA is capable of stimulating amino acid incorporation in the in vitro translation system, however, it does not direct the synthesis of LHCP.
Similar content being viewed by others
Abbreviations
- LHCP:
-
light-harvesting chlorophyll a/b protein
- PAL:
-
phenylalanine ammonia-lyase
References
Apel, K. (1979) Phytochrome-induced appearance of mRNA activity for the apoprotein of the light-harvesting chlorophyll a/b protein of barley (Hordeum vulgare). Eur. J. Biochem.97, 183–188
Apel, K., Kloppstech, K., (1978) The plastid membranes of barley (Hordeum vulgare). Light-induced appearance of the mRNA coding for the apoprotein of the light-harvesting chlorophyll a/b protein. Eur. J. Biochem.85, 581–588
Armond, P.A., Staehelin, L.A., Arntzen, C.J. (1977) Spatial relationship of photosystem I, photosystem II and the light-harvesting complex in chloroplast membranes. J. Cell Biol.73, 400–418
Bogorad, L. (1967) In: Organization biosynthesis, pp. 395–418, Vogel, A.J., Lampen, J.O., Bryson, V., eds., Academic Press, New York
Bonner, W.M., Laskey, R.A. (1974) A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur. J. Biochem.46, 83–88
Chua, N.H., Blomberg, F. (1979) Immunochemical studies of thylakoid membrane polypeptides from spinach andChlamydomonas reinhardtii. J. Biol. Chem.254, 215–223
Chua, N.H., Matlin, K., Bennoun, P. (1975) A chlorophyll-protein complex lacking in photosystem I mutants ofChlamydomonas reinhardtii. J. Cell Biol.67, 361–377
Kloppstech, K., Schweiger, H.G. (1976) In vitro translation of poly(A)RNA fromAcetabularia. Cytobiologie13, 394–400
Meyer, T.S., Lamberts, B.L. (1965) Use of Coomassie brilliant blue R 250 for the electrophoresis of microgram quantities of parotid saliva proteins on acrylamide-gel strips. Biochim. Biophys. Acta107, 144–145
Müller, M., Viro, M., Balke Ch., Kloppstech, K. (1979) Light-induced appearance of the rRNA for a chloroplast membrane protein in polysomes. Its in vitro translation and processing of the precursor protein. Hoppe Seyler's Z. Physiol. Chem.360, 330
Neville, D.M. (1971) Molecular weight determination of proteindodecylsulfate complexes by gel electrophoresis in a discontinous buffer system. J. Biol. Chem.246, 6328–6334
Roberts, B.E., Paterson, B.M. (1970) Efficient translation of tobacco mosaic virus RNA and globin 9 S RNA in a cell-free system from commercial wheat germ. Proc. Natl. Acad. Sci. USA70, 2330–2334
Schröder, J., Kreuzaler, F., Schäfer, E., Hahlbrock, K. (1979) Concomitant induction of phenylalanine ammonia-lyase and flavanone synthase mRNAs in irradiated plant cells. J. Biol. Chem.254, 57–65
Seitz, U., Richter, G. (1970) Isolierung und Charakterisierung schnell markierter, hochmolekularer RNS aus frei suspendierten Calluszellen der Petersilie (Petroselinum sativum). Planta92, 309–326
Spieß, E., Seitz, U. (1975) Quantitative determination of growth-dependent changes in the ultrastructure of freely suspended callus cells of parsley (Petroselium crispum). Ber. Dtsch Bot. Ges.88, 319–328
Takebe, J., Labib, G., Melchers, G. (1971) Regeneration of whole plants from isolated mesophyll protoplasts from tobacco. Naturwissenschaften58, 318–320
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Müller, M., Viro, M., Balke, C. et al. Polyadenylated mRNA for the light-harvesting chlorophyll a/b protein. Planta 148, 444–447 (1980). https://doi.org/10.1007/BF02395312
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02395312