Abstract
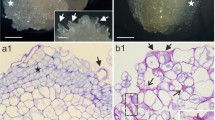
Carrot (Daucus carota L.) embryogenic callus (EC) loses its embryogenic competence and becomes nonembryogenic callus (NC) during long-term culture. With the loss of embryogenic competence, the cell clusters become smaller and the extent of intercellular attachments is reduced. Pectic fractions prepared from EC and NC were separated into two subfractions by gel filtration. A difference in sugar composition between EC and NC was found only in the high-molecular-mass (ca. 1300 kDa) subfraction, and the ratio of the amount of arabinose to that of galactose (Ara/Gal) was strongly and positively correlated with the size of cell clusters in several different cultures. From the results of sugar-composition and methylation analyses, and the results of treatment with exo-arabinanase, models of the neutral sugar chains of pectins from EC and NC are proposed. Both neutral sugar chains are composed of three regions. The basal region is composed of linearly linked arabinan 5-Araf> moieties in both types of callus. The middle galactan region is composed of 6-linked galactose, some of which branches at the 3 and 4 positions, and this region is larger and more frequently branched in NC than in EC. Finally, the terminal arabinan region is composed of 5-linked arabinose, branched at the 3 position, and the size of the terminal arabinan is larger in EC than in NC. The significance of the neutral sugar chains of pectins in the interaction of cell wall components and intercellular attachment is discussed.
Similar content being viewed by others
Abbreviations
- Ara/Gal:
-
ratio (w/w) of the amount of arabinose to that of galactose
- EC:
-
embryogenic callus
- NC:
-
non-embryogenic callus
- T-Araf :
-
terminal arabinose
References
Albersheim P, Nevins DJ, English PD, Karr A (1967) A method for the analysis of sugars in plant cell-wall polysaccharides by gasliquid chromatography. Carbohydr Res 5: 340–345
Bonner J (1950a) The Pectic Substances. In: Plant biochemistry. Academic Press, New York, pp 99–108
Dawson DM, Melton LD, Watkins CB (1992) Cell wall changes in nectarines (Prunus persica). Solubilization and depolymerization of pectic and neutral polymers during ripening and in mealy fruit. Plant Physiol 100: 1203–1210
Dopico B, Labrador E, Nicolas G (1986) Characterization and localization of the cell walls autolysis substrate in Pisum sativum epicotyls. Plant Sci 44: 155–161
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28: 350–356
Fincher GB, Stone BA (1981) Metabolism of noncellulosic polysaccharides. In: Plant carbohydrate. II. Extracellular carbohydrates. Springer-Verlag, Berlin, pp 68–132
Fry SC (1983) Feruloylated pectins from the primary cell wall: their structure and possible functions. Planta 157: 111–123
Galambos JT, McCain JR (1967) The reaction of carbazole with carbohydrate. I. Effect of borate and sulfamate on the carbazole color of sugar. Anal Biochem 19: 119–143
Goldberg R, Morvan C, Herve du Penhoat C, Michon V (1989a) Structure and properties of acidic polysaccharides from mung bean hypocotyls. Plant Cell Physiol 30: 163–173
Hakomori S (1964) A rapid permethylation of glycolipid, and polysaccharide catalyzed by methylsulfinyl carbonion in dimethyl sulfoxide. J Biochem 55: 205–208
Ishii T (1991) Isolation and characterization of a diferuloyl arabinoxylan hexasaccharide from bamboo shoot cell-walls. Carbohydr Res 219: 15–22
Kikuchi A, Satoh S, Nakamura N, Fujii T (1995) Differences in pectic polysaccharides between carrot embryogenic and nonembryogenic calli. Plant Cell Rep 14: 279–284
Kiyosue T, Satoh S, Kamada H, Harada H (1993) Somatic embryogenesis in higher plant. J Plant Res Spec Iss 3: 75–82
Kobayashi H, Fukuda H (1994) Involvement of calmodulin and calmodulin-binding proteins in the differentiation of tracheary elements in Zinnia cells. Planta 194: 388–394
Labrador E, Nicolas G (1985) Autolysis of cell walls in pea epicotyls during growth. Enzymatic activities involved. Physiol Plant 64: 541–546
Liners F, Van Cutsem P (1992) Distribution of pectic polysaccharides throughout walls of suspension-cultured carrot cells. An immunocytochemical study. Protoplasma 170: 10–21
Massiot P, Rouau X, Thibault JF (1988) Characterization of the extractable pectins and hemicelluloses of the cell wall of carrot. Carbohydr Res 172: 229–242
Mu~noz FJ, Dopico B, Labrador E (1993) Effect of osmotic stress on the growth of epicotyls of Cicer arietinum in relation to changes in cell wall composition. Physiol Plant 87: 552–560
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15: 473–497
Nevins DJ, English PD, Albersheim P (1968) Changes in cell wall polysaccharides associated with growth. Plant Physiol 43: 914–922
Nishitani K, Masuda Y (1979) Growth and cell wall changes in azuki bean epicotyls. I. Changes in wall polysaccharides during intact growth. Plant Cell Physiol 20: 63–74
Nishitani K, Masuda Y (1982) Roles of auxin and gibberellic acid in growth and maturation of epicotyls of Vigna angularis: Cell wall changes. Physiol Plant 56: 38–45
Redgwell JR, Melton LD, Brasch DJ (1992) Cell wall dissolution in ripening kiwifruit (Actinidia deliciosa). Solubilization of the pectic polymers. Plant Physiol 98: 71–81
Reinert J (1959) Uber die Kontrolle der Morphogenese und die Induktion von Adventivembryonen an Gewebenkulturen aus Karotten. Planta 53: 318–333
Reinert D, Backs-Husemann, Zerban H (1970) Determination of embryo and root formation in tissue Les Cultures de Tissue de Plantes. CNRS, Paris, pp 261–268
Revilla G, Victoria M, Zarra I (1986) Cell wall autolysis in Cicer arietinum L. epicotyls. J Plant Physiol 122: 147–157
Rushing JW, Huber DJ (1987) Effect of NaCl, pH and Ca2+ on autolysis of ioslated tomato fruit cell walls. Physiol Plant 70: 78–84
Sakurai N, Tanaka T, Kuraishi S (1987) Changes in wall polysaccharides of squash (Cucurbita maxima Duch.) hypocotyls under water stress condition. I. Wall sugar composition and growth as affected by water stress. Plant Cell Physiol 28: 1051–1058
Sandford PA, Conrad HE (1966) The structure of Acrobacter aerogenes A3 (S1) p.s. I. A reexamination using improved procedures for methylation analysis. Biochemistry 5: 1508–1516
Satoh S, Kamada H, Harada H, Fujii T (1986) Auxin-controlled glycoprotein release into the medium of embryogenic carrot cells. Plant Physiol 81: 931–933
Shibuya N (1984) Phenolic acids and their carbohydrate esters in rice endosperm cell walls. Phytochemistry 23: 2233–2237
Stoddart RW, Barrett AJ, Northcote DH (1967) Pectic polysaccharides of growing plant tissues. Biochem J 102: 194–204
Tanimoto E (1988) Gibberellin regulation of root growth with change in galactose contents of cell walls in Pisum sativum. Plant Cell Physiol 29: 269–280
Uesaka E, Sato M, Raiju M, Kaji A (1978) α-l-Arabinofuranosidase from Rhodotorula flava. J Bacteriol 133: 1073–1077
York WS, Darvill AG, McNeil M, Stevenson TT, Albershein P (1985) Isolation and characterization of plant cell wall and cell wall components. Methods Enzymol 118: 3–40
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors are grateful to Dr. Naoto Shibuya of the National Institute of Agrobiological Resources for his gift of exo-arabinanase.
Rights and permissions
About this article
Cite this article
Kikuchi, A., Edashige, Y., Ishii, T. et al. Variations in the structure of neutral sugar chains in the pectic polysaccharides of morphologically different carrot calli and correlations with the size of cell clusters. Planta 198, 634–639 (1996). https://doi.org/10.1007/BF00262652
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00262652