Summary
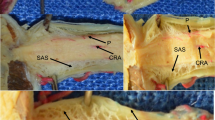
An ultrastructural and tracer study was undertaken to determine normal outflow pathways of cerebrospinal fluid (CSF) at the terminal subarachnoid space (SAS) of the optic nerve. In the morphological studies, the optic nerve dura and arachnoid were found to be continuous with the sclera of the eye beyond the optic nerve SAS. The pia mater is continuous with the inner sciera and the lamina fusca of the eye. Montages and serial sections demonstrated that the distal SAS is divided into numerous tortuous channels to form an “arachnoidal trabecular meshwork”. Spaces of this meshwork continue into “microcanals” which bypass the outer arachnoid barrier layers of the optic nerve meninges to reach the sclera and posterior intraorbital connective tissue. Ferritin infused into the cisterna magna entered the optic nerve SAS within 1 min and reached arachnoidal trabecular meshwork channels and the microcanals within 8 min. It then passed into intraorbital connective tissue spaces at the posterior pole of the eye. Ferritin appeared to be blocked by the lamina fusca and a newly discovered “posterior compact zone” which together prevented its entrance into the choroidal interstitium. These observations suggest that a “ subarachnoidal-scleral-orbital outflow pathway” provides a route for CSF drainage from the optic nerve SAS to intraorbital connective tissue. The previously described “posterior uveal compartment” in the hamster eye (Kelly et al. 1983) appears to be relatively isolated from this subarachnoidal-scleral-orbital CSF outflow.
Similar content being viewed by others
References
Anderson DR, Hoyt WF (1969) Ultrastructure of intraocular portion of human and monkey optic nerve. Arch Ophthalmol 82:506–530
Bill A (1977) Basic physiology of the drainage of aqueous humor. Exp Eye Res 25 Suppl: 291–304
Bill A (1980) Drainage of intraocular fluids. In: Jose G Cunha-Vaz (ed) The blood retinal barriers. Plenum Corp New York, pp 179–193
Bradbury MWB, Cole DF (1980) The role of the lymphatic system in drainage of cerebrospinal fluid and aqueous humor. J Physiol (Lond) 299:353–365
Bradbury MWB, Westrop RJ (1984) Lymphatics and the drainage of cerebrospinal fluid. In Shapiro K, Marmarou A, Portnoy H (eds) Hydrocephalus. Raven press, New York, pp 69–81
Brierley JB, Field EJ (1948) The connexions of the spinal subarachnoid with the lymphatic system. J Anat 82:153–166
Brierley JB, Field EJ, Yoffey JM (1949) Passage of indian ink particles from the cranial subarachnoid space. J Anat (Lond) 83:77–78
Cole DF, Monro PAG (1976) The use of fluorescein-labeled dextrans in investigation of aqueous humor outflow in the rabbit. Exp Eye Res 23:571–585
Faber WM (1937) The nasal mucosa and the subarachnoid space. Am J Anat 62:121–148
Field EJ, Brierley JB (1949) The retro-orbital tissues as a site of outflow of cerebrospinal fluid. Proc R Soc Med 42:447–450
Frazier CH, Peet MM (1914) Factors of influence in the origin and circulation of the cerebrospinal fluid. Am J Physiol 35:268–282
Hageman GS, Kelly DE (1984) Fibrillar and cytoskeletal substructure of tight junctions: analysis of single-stranded tight junctions linking fibroblasts of the lamina fusca in hamster eyes. Cell Tissue Res 238:545–557
Inomata H, Bill A, Smelser GK (1972) Aqueous humor pathways through the trabecular meshwork and into Schlemm's canal in the cynomolgus monkey (Macaco irus), an electron microscopic study. Am J Ophthalmol 73:760–789
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137A-138A
Kelly DE, Hageman GS, McGregor JA (1983) Uveal compartmentalization in the hamster eye revealed by fine structural and tracer studies: implications for uveoscleral outflow. Invest Ophthalmol Vis Sci 24:1288–1304
McComb JG, Davson H, Hyman S, Weiss MH (1982) Cerebrospinal fluid drainage as influenced by ventricular pressure in the rabbit. J Neurosurg 56:790–797
McComb JG, Hyman S, Weiss MH (1984) Lymphatic drainage of cerebrospinal fluid in the cat. In Shapiro K, Marmarou A, Portnoy H (eds) Hydrocephalus. Raven press, New York, pp 83–98
Mollenhauer HH (1964) Plastic embedding mixtures for use in electron microscopy. Stain Tech 39:111–114
Nabeshima S, Reese TS, Landis DMD, Brightman MW (1975) Junctions in the meninges and marginal glia. J Comp Neurol 164:127–170
Pino RM, Essner E (1980) Structure and permeability to ferritin of the choriocapillary endothelium of the rat eye. Cell Tissue Res 208:21–27
Richardson KC, Jarett L, Finkle EH (1960) Embedding in epoxy resins for ultra-thin sectioning in electron microscopy. Stain Technol 35:313–325
Shen JY (1984) CSF outflow pathways at the optic nerve-eye junction in the hamster. Anat Rec 208:164A
Sherman SH, Green K, Laties AM (1978) The fate of anterior chamber fluorescein in the monkey eye. 1. The anterior chamber outflow pathways. Exp Eye Res 27:159–173
Tripathi RC (1977) The functional morphology of the outflow systems of ocular and cerebrospinal fluids. Exp Eye Res 25 suppl:65–116
Weed LH (1914) Studies on cerebro-spinal fluid. No. 111. The pathways of escape from the subarachnoid spaces with particular references to the arachnoid villi. J Med Res 31:51–91
Weed LH (1917) An anatomical consideration of the cerebrospinal fluid. Anat Rec 12:461–496
Wegefarth P (1914) Studies on cerebro-spinal fluid. No VI The establishment of drainage of intra-ocular and intracranial fluids into the venous system. J Med Res 31:149–166
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shen, JY., Kelly, D.E., Hyman, S. et al. Intraorbital cerebrospinal fluid outflow and the posterior uveal compartment of the hamster eye. Cell Tissue Res. 240, 77–87 (1985). https://doi.org/10.1007/BF00217560
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00217560