Summary
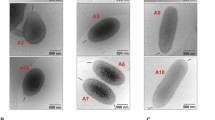
It has been suggested that orthogonal arrays of particles may increase the rigidity of plasma membrane, as does cholesterol. Therefore, using freeze-fractured non- pigmented ciliary epithelium, the distribution of such arrays was compared to the distribution of membrane deformations induced by the sterol-probes filipin and digitonin in different domains of the basolateral plasma membrane. The distribution of orthogonal arrays of particles was homogeneous between different regions of the basolateral membrane of the non-pigmented ciliary epithelium, while the number of filipin-induced alterations was nearly 4 times higher in the membrane domains not in contact with the basal lamina than in domains in contact with it. Contrary to the homogeneous distribution of arrays, digitonin-induced deformations also differed markedly in these two basolateral membrane domains. Considering that a marked positive response to sterol probes implies a high sterol content, we conclude that orthogonal arrays of particles can occur in plasma membrane regions well-provided with cholesterol and not in direct contact with the basal lamina. Other possible roles of these arrays are discussed.
Similar content being viewed by others
References
Bartels H, Miragall F (1986) Orthogonal arrays of particles in the plasma membrane of pneumocytes. J Submicrosc Cytol 18:637–646
Behnke O, Tranum-Jensen J, Deurs B van (1984) Filipin as a cholesterol probe. II. Filipin-cholesterol interaction in red blood cell membranes. Eur J Cell Biol 35:200–215
Bordi C, Amherdt M, Perrelet A (1986) Orthogonal arrays of particles in the gastric parietal cell of the rat: differences between superficial and basal cells in the gland and after pentagastrin or metiamide treatment. Anat Rec 215:28–34
Brown D, Montesano R, Orci L (1982) Patterns of filipin-sterol complex distribution in intact erythrocytes and intramembrane particle-aggregated ghost membranes. J Histochem Cytochem 30:702–706
Caldwell RB (1987) Filipin and digitonin studies of cell membrane changes during junction breakdown in the dystrophic rat retinal pigment epithelium. Curr Eye Res 6:515–526
Costello MJ, McIntosh TJ, Robertson JD (1985) Membrane specializations in mammalian lens fiber cells: distribution of square arrays. Curr Eye Res 4:515–526
Demel RA, De Kruyff B (1976) The function of sterols in membranes. Biochim Biophys Acta 457:109–132
Dermietzel R (1974) Junctions in the central nervous system of the cat. III. Gap junctions and membrane-associated orthogonal particle complexes (MOPC) in astrocytic membranes. Cell Tissue Res 149:121–135
Elias PM, Friend DS, Goerkc J (1979) Membrane sterol heterogeneity. Freeze-fracture detection with saponins and filipin. J Histochem Cytochem 27:1247–1260
Ellisman MH, Rash JE, Staehelin LA, Porter KR (1976) Studies of excitable membranes. II. A comparison of specializations at neuromuscular junctions and nonjunctional sarcolemmas of mammalian fast and slow twitch muscle fibers. J Cell Biol 68:752–774
Feltkamp CA, Waerden AWM van der (1982) Membrane-associated proteins affect the formation of filipin-cholesterol complexes in viral membranes. Exp Cell Res 140:289–297
Fischbeck KH, Bonilla E, Schotland DL (1982) Freeze-fracture analysis of plasma membrane cholesterol in fast and slow twitch muscles. J Ultrastruct Res 81:117–123
Friend DS, Bearer EL (1981) β-Hydroxysterol distribution as determined by freeze-fracture cytochemistry. Histochem J 13:535–546
Gordon RE (1985) Orthogonal arrays in normal and injured respiratory airway epithelium. J Cell Biol 100:648–651
Gotow T (1984) Cytochemical characteristics of astrocytic plasma membranes specialized with numerous orthogonal arrays. J Neurocytol 13:431–448
Gotow T, Yoshikawa H, Hashimoto PH (1985) Distribution patterns of orthogonal arrays and alkaline phosphatase in plasma membranes of satellite cells in rat spinal ganglia. Anat Embryol 171:171–179
Hatton JD, Ellisman MH (1981) The distribution of orthogonal arrays and their relationship to intercellular junctions in neuroglia of freeze-fractured hypothalamo-neurohypophysial system. Cell Tissue Res 215:309–323
Hirsch M (1985) Presence of orthogonal arrays of particles in the ciliary body epithelium of the rat. Cell Biol Int Rep 9:867
Humbert F, Pricam C, Perrelet A, Orci L (1975) Specific plasma membrane differentiations in the cells of the kidney collecting tubule. J Ultrastruct Res 52:13–20
Jain MK (1975) Role of cholesterol in biomembranes and related systems. Curr Top Membr Transp 6:1–57
Karnovsky MJ (1982) Intramembranous cytochemistry: a perspective. Lab Invest 46:637–639
Kinsky SC, Luse SA, Van Deenan LLM (1966) Interaction of polyene antibiotics with natural and artificial membrane systems. Fed Proc 25:1503–1510
Landis DMD, Reese TS (1974) Arrays of particles in freeze-fractured astrocytic membranes. J Cell Biol 60:316–320
Landis DMD, Reese TS (1981) Membrane structure in mammalian astrocytes: a review of freeze-fracture studies on adult, developing, reactive and cultured astrocytes. J Exp Biol 95:35–48
Lange Y, Slayton JM (1982) Interaction of cholesterol and lysophosphatidylcholine in determining red cell shape. J Lipid Res 23:1121–1127
Lo W-K, Harding CV (1984) Square arrays and their role in ridge formation in human lens fibers. J Ultrastruct Res 86:228–245
Orci L, Singh A, Amherdt M, Brown D, Perrelet A (1981) Microheterogeneity of protein and sterol content in kidney podocyte membrane. Nature 293:646–647
Rash JE, Staehelin LA, Ellisman MH (1974) Rectangular arrays of particles on freeze-cleaved membranes are not gap junctions. Exp Cell Res 86:187–190
Schmalbruch H (1985) Skeletal muscle. In: Oksche A, Vollrath L (eds) Handbook of microscopic anatomy. Springer, Berlin Heidelberg New York: vol II/6, pp 137–138
Severs NJ, Robenek H (1983) Detection of microdomains in biomembranes. Biochim Biophys Acta 737:373–408
Severs NJ, Warren RC, Barnes SH (1981) Analysis of membrane structure in the transitional epithelium of rat urinary bladder. 3. Localization of cholesterol using filipin and digitonin. J Ultrastruct Res 77:160–188
Simons HL, Severs NJ (1986) Plasmalemmal vesicles and the effects of sterol-binding agents in rabbit aortic endothelium. J Cell Sci 83:141–153
Steer CJ, Bisher M, Blumenthal R, Steven AC (1984) Detection of membrane cholesterol by filipin in isolated rat liver coated vesicles is dependent upon removal of the clathrin coat. J Cell Biol 99:315–319
Streeten BW (1982) Ciliary body. In: Jacobiec FA (ed) Ocular anatomy, embryology and teratology. Harper and Row, Philadelphia, pp 303–330
Tamm SL, Tamm S (1983) Distribution of sterol-specific complexes in a continually shearing region of a plasma membrane and at procaryotic-eucaryotic cell junctions. J Cell Biol 97:1098–1106
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hirsch, M., Gache, D. & Noske, W. Orthogonal arrays of particles in non-pigmented cells of rat ciliary epithelium: Relation to distribution of filipin- and digitonin-induced alterations of the basolateral membrane. Cell Tissue Res. 252, 165–173 (1988). https://doi.org/10.1007/BF00213838
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00213838