Abstract
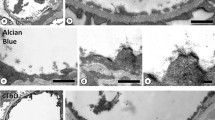
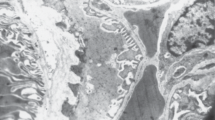
Scanning and transmission electron microscopy were used to study the ultrastructural characteristics and positions of granulated peripolar cells in newborn lamb kidney. Following tissue fixation by vascular perfusion in situ, the vascular pole region of the glomerulus was exposed for examination by scanning electron micoscopy following removal of the glomerular tuft. Peripolar cells were recognized by their surface morphology enabling their quantification and an assessment of the relationship of their position in the renal cortex. The prominent expression of peripolar cells in this species was confirmed. Almost every vascular pole examined revealed peripolar cells (405 out of 407; 99.5%) and thus, throughout the cortex, the distribution of peripolar cells was the same as the distribution of renal corpuscles. Larger, more protruding peripolar cells were observed in the outer cortical renal corpuscles. The numbers of peripolar cells encircling each vascular pole ranged from 1 to 10. There was no correlation between number of granulated peripolar cells at the vascular pole and the position of the renal corpuscle within the renal cortex. As viewed by transmission electron microscopy, organelles of protein synthesis were abundant in the cytoplasm of peripolar cells. Exocytosis of cytoplasmic granules was observed by both scanning and transmission electron microscopy implying that a process of regulative secretion occurs from these cells. The use of ultrastrural techniques has provided evidence supporting the concept that peripolar cells are prominent in the cuff region of each renal corpuscle of the newborn lamb and further-more that peripolar cells in this species most likely have a secretory function.
Similar content being viewed by others
References
Alcorn D, Cheshire GR, Coghlan JP, Ryan GB (1984) Peripolar cell hypertrophy in the renal juxtaglomerular region of newborn sheep. Cell Tissue Res 222:101–111
Ballard PC (1979) Glucocorticoids and differentiation. In: Baxter JD, Rousseau GG (eds) Glucocorticoid hormone action. Springer, Berlin. pp 493–515
Casellas D (1986) A method for scanning electron microscopic observation of glomerular vascular poles in rat kidneys. J Electron Microse Tech 4:63–64
Downie I, Gardiner DS, Downie TT, Gibson IW, Kenyon C, More IAR, Lindop GBM (1992) Non-granulated peripolar cells exist in the rat glomerulus. Cell Tissue Res 268:567–570
Ermak TH, Rothman SS (1980) Large decrease in zymogen granule size in the postnatal rat pancreas. J Ultrastruct Mol Struct Res 70:242–256
Farquhar MG (1985) Progress in unravelling pathways of Golgi traffic. Ann Rev Cell Biol 1:447–488
Frank M, Kriz W (1982) Scanning electron microscopy studies of the vascular pole of the rat glomerulus. Anat Rec 204:149–152
Gall JAM, Alcorn D, Butkus A, Coghlan JP, Ryan GB (1986) Distribution of glomerular peripolar cells in different mammalian species. Cell Tissue Res 24:203–208
Gardiner DS, Lindop GBM (1985) The granular peripolar cell of the human glomerulus: a new component of the juxtaglomerular apparatus? Histopathology 9:675–685
Gardiner DS, Lindop GBM (1992) The glomerular peripolar cell —an immunohistochemical study. APMIS 100:107–115
Gardiner DS, More IAR, Lindop GBM (1986) The granular peripolar cell of the human glomerulus: an ultrastructural study. J Anat 146:31–43
Gibson IW, More IAR, Lindop GBM (1989) A scanning electron microscopic study of the peripolar cell of the rat renal glomerulus. Cell Tissue Res 257:201–206
Hanner RH, Ryan GB (1980) Ultrastructure of the renal juxtaglomerular complex and peripolar cells in the axolotl (Ambystoma mexicanum) and toad (Bufo marinus). J Anat 130:445–455
Hill PA, Coghlan JP, Scoggins BA, Ryan GB (1983) Ultrastructural changes in the sheep renal juxtaglomerular apparatus in response to sodium depletion or loading. Pathology 15:463–473
Hollywell CA, Jaworowski A, Thumwood C, Alcorn D, Ryan GB (1992) Immunohistochemical localization of transthyretin in glomerular peripolar cells of newborn sheep. Cell Tissue Res 267:193–197
Kelly G, Downie I, Gardiner DS, More IAR, Lindop GBM (1990) The peripolar cell: a distinctive cell type in the mammalian glomerulus. Morphological evidence from a study of sheep. J Anat 168:217–227
Kelly RB (1985) Pathways of protein secretion in eukaryotes. Science 230:25–32
Kleinman LI (1978) The Kidney. In: Stave U (ed) Perinatal physiology. Plenum Press, New York London, pp 589–616
Kovalchuk LE (1987) Ultrastructure of the juxtaglomerular complex of the kidney and peripolar cells in the sand lizard. Arkh Anat Gistol Embriol 93:93–98
Lacy ER, Reale E (1989) Granulated peripolar epithelial cells in the renal corpuscle of marine elasmobranch fish. Cell Tissue Res 257:61–67
Lendrum AC, Fraser DS, Slidders W, Henderson R (1962) Studies on the character and staining of fibrin. J Clin Pathol 15:401–413
Mbassa GK (1989) Peripolar cells form the majority of granulated cells in the kidneys of antelopes and goats. Acta Anat (Basel) 135:158–163
McDougall EI (1965) Proteinuria of suckling ruminants. Biochem J 94:101–105
Morild I, Christensen JA, Mikeler E, Bohle A (1988) Peripolar cells in the avian kidney. Virchows Archiv [A] 412:471–477
Pratt RE, Carleton JE, Richie JP, Heussen C, Dzau VJ (1987) Human renin biosynthesis and secretion in normal and ischaemic kidneys. Proc Natl Acad Sci USA 84:7837–7840
Redman RJ, Sreebny LM (1971) Morphologic and biochemical observations on the development of the rat parotid gland. Dev Biol 25:248–279
Ryan GB, Coghlan JP, Scoggins BA (1979) The granulated peripolar cell: a potential secretory component of the renal juxtaglomerular complex. Nature 277:655–656
Ryan GB, Alcorn D, Coghlan JP, Hill PA, Jacobs R (1982) Ultrastructural morphology of granule release from juxtaglomerular myoepithelioid and peripolar cells. Kidney Int 22 [Supp 12]: S3-S8
Spitzer A (1982) The role of the kidney in sodium homeostasis during maturation. Kidney Int 21:539–545
Taugner R, Hackenthal E (1989) The juxtaglomerular apparatus, 1st edn. Springer, Berlin Heidelberg New York, pp 50–52
Trahair JF, Ryan GB (1988) Immunohistochemical identification of plasma proteins in cytoplasmic granules of peripolar cells in the sheep. J Anat 160:109–115
Trahair JF, Ryan GB (1989) Co-localization of neuron-specific enolase-like and kallikrein-like immunoreactivity in ductal and tubular epithelium of sheep salivary gland and kidney. J Histochem Cytochem 37:309–314
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thumwood, C.M., McCausland, J., Alcorn, D. et al. Scanning and transmission electron-microscopic study of peripolar cells in the newborn lamb kidney. Cell Tissue Res 274, 597–604 (1993). https://doi.org/10.1007/BF00314558
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00314558