Summary
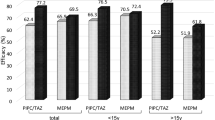
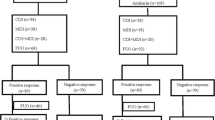
In a nonblind, randomised, parallel-group study, initial empirical monotherapy with meropenem 1 g intravenously every 8 h was compared to an identical dosage of imipenem/cilastatin for the treatment of 66 febrile episodes in 61 adult neutropenic patients. 25/31 episodes treated with meropenem and 24/30 imipenem/cilastatin-treated episodes were still receiving unmodified therapy at 72 h (primary endpoint); this difference was not statistically significant. By the end of the treatment courses, 18/31 meropenem-treated episodes had responded clinically (cured or improved) compared with 18/30 episodes treated with imipenem/cilastatin. Another ten episodes initially treated with meropenem and six episodes treated with imipenem/cilastatin were cured after an additional antimicrobial agent had been administered (cured with modification). Satisfactory bacteriological responses (eradication plus presumed eradication) at the end of unmodified therapy was 9/11 in the meropenem group and 14/16 in the comparator group. Both regimes were well tolerated; however, there were more reports of nausea and/or vomiting in the imipenem/cilastatin group (7/33 vs. 2/33 in the meropenem group). The carbapenems meropenem and imipenem/cilastatin appear to be suitable agents for empirical monotherapy of febrile episodes in neutropenic patients. Meropenem may be better tolerated than imipenem/cilastatin, allowing optimal dosing in this patient population.
Zusammenfassung
In einer offenen, randomisierten Vergleichsstudie wurde die initiale empirische Monotherapie mit Meropenem in einer Dosierung von 3 × 1g täglich verglichen mit Imipenem/Cilastatin in der gleichen Dosierung bei 66 Fieberepisoden von 61 erwachsenen neutropenischen Patienten. 72 Stunden nach Therapiebeginn erhielten noch 25/31 Patienten aus der Meropenem-Gruppe und 24/30 Patienten aus der Behandlungsgruppe mit Imipenem/Cilastatin die usprünglich zugewiesene Initialtherapie (Hauptzielkriterium). Bei Therapieende waren in der Meropenem-Gruppe 18/31 Episoden geheilt oder gebessert gegenüber 18/30 in der Gruppe mit Imipenem/Cilastatin. Weitere 10 Infektionsepisoden, die initial mit Meropenem behandelt wurden, und sechs Infektionsepisoden aus der Vergleichsgruppe konnten erfolgreich behandelt werden, nachdem eine zusätzliche antimikrobielle Substanz verabreicht worden war (Heilung nach Therapiemodifikation). Das zufriedenstellende bakteriologische Ansprechen (Keimelimination sowie vermutete Keimelimination) mit der Initialtherapie Meropenem betrug 9/11 und 14/16 mit Imipenem/Cilastatin. Beide Antibiotika wurden gut vertragen; es wurden jedoch unter Imipenem/Cilastatin mehr Fälle von Übelkeit und/oder Erbrechen registriert (7/33 vs 2/33 unter Meropenem). Die Carbapenem-Antibiotika Meropenem und Imipenem/Cilastatin scheinen geeignet für die empirische Monotherapie bei Fieberepisoden neutropenischer Patienten zu sein. Meropenem erlaubt durch seine offensichtlich bessere Verträglichkeit im Vergleich zu Impienem/Cilastatin auch eine optimale Dosierung bei diesem Patientengut.
Similar content being viewed by others
References
De Pauw, B. E., Kauw, F., Muytijens, H., Williams, K. J., Bothof, T. Randomized study of ceftazidime versus gentamicin plus cefotaxime for infections in severe granulocytopenic patients. J. Antimicrob. Chemother. 12 (Suppl. A) (1983) 93–99.
De Pauw, B., Williams, K., de Neeff, J., Bothof, T., de Witte, T., Holdrinet, R., Haanen, C. A randomized prospective study of ceftazidime versus ceftazidime plus flucloxacillin in the empiric treatment of febrile episodes in severely neutropenic patients. Antimicrob. Agents Chemother. 28 (1985) 824–828.
Pizzo, P. A., Hathorn, J. W., Hiemenz, J., Browne, M., Commers, J., Cotton, D., Gress, J., Longo, D., Marshall, D., McKnight, J., Rubin, M., Skelton, J., Thaler, M., Wesley, R. A randomized trial comparing ceftazidime alone with combination antibiotic therapy in cancer patients with fever and neutropenia. N. Engl. J. Med. 315 (1986) 552–558.
Shah, P. Resistenzsituation im klinischen Alltag. Arzneimitteltherapie 3 (1992) 94–95.
Bauernfeind, A., Rosenthal, E. J. K., Eberlein, E., Holley, M., Schweighart, S. Spread ofKlebsiella pneumoniae producing SHV-5 beta-lactamase among hospitalized patients. Infection 21 (1993) 18–22.
Edwards, J. R., Turner, P. J. Laboratory data which differentiate meropenem and imipenem. Scand. J. Infect. Dis. (Suppl. 96) (1995) 5–10.
Liang, R., Yung, R., Chiu, E., Chau, P.-Y., Chan, T.-K., Lam, W.-K., Todd, D. Ceftazidime versus imipenem-cilastatin as initial monotherapy for febrile neutropenic patients. Antimicrob. Agents Chemother. 34 (1990) 1336–1341.
Freifeld, A. G., Walsh, T., Marshall, D., Gress, J., Steinberg, S. M., Hathorn, J., Rubin, M., Jarosinski, P., Gill, V., Young, R. C., Pizzo, P. A. Monotherapy for fever and neutropenia in cancer patients: a randomized comparison of ceftazidime versus imipenem. J. Clin. Oncol. 13 (1995) 165–176.
The Meropenem Study Group of Leuven, London and Nijmegen Equivalent efficacies of meropenem and ceftazidime as empirical monotherapy of febrile, neutropenic patients. J. Antimicrob. Chemother. 36 (1995) 185–200.
Cometta, A. forthe International Antimicrobial Therapy Cooperative Group of the EORTC and the GIMEMA Infection Program Monotherapy with meropenem versus combination therapy with ceftazidime plus amikacin as empiric therapy for fever in granulocytopenic patients with cancer. Antimicrob. Agents Chemother. 40 (1996) 1108–1115.
Winston, D. J., Ho, W. G., Bruckner, D. A., Champlin, R. E. Beta-lactam antibiotic therapy in febrile granulocytopenic patients. A randomized trial comparing cefoperazone plus piperacillin, ceftazidime plus piperacillin, and imipenem alone. Ann. Intern. Med. 115 (1991) 849–859.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shah, P.M., Stille, W., Böhme, A. et al. Empirical monotherapy with meropenem versus imipenem/cilastatin for febrile episodes in neutropenic patients. Infection 24, 480–484 (1996). https://doi.org/10.1007/BF01713054
Issue Date:
DOI: https://doi.org/10.1007/BF01713054