Abstract
We made a comparative clinical, immunopathological and therapeutic evaluation in 17 patients with polymyositis (PM) and 12 patients with dermatomyositis (DM), followed up at our Neuromuscular Center. DM can be distinguished by its clinical appearance and pathological changes. Current evidence suggests that it results from vasculopathy.
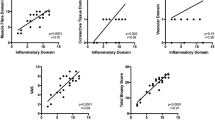
For studying these inflammatory myopathies we used multifactorial diagnostic criteria, evaluating the therapeutic response by means of a composite clinical and functional score in a longitudinal study. In muscle biopsy specimens we characterized with monoclonal antibodies T lymphocyte subpopulations (CD4, CD8), macrophages, IgG, IgM, C1q, C3, C4 complement fractions, MHC-I, MHC-II. In PM the cell-mediated immunity was more pronounced and in sowithin muscle fibers. Our patients were treated with steroids; in resistant cases azathioprine, cyclophosphamide, plasmapheresis, high-dose intravenous immunoglobulins (ivIgG) and total body irradiation were added to the therapeutic schedule.
Sommario
È stata condotta una valutazione comparativa di tipo clinico, immunopatologico e terapeutico di 17 pazienti affetti da Polimiosite (PM) e 12 affetti da Dermatomiosite (DM), seguiti presso il nostro Centro.
Nello studio di queste miopatie infiammatorie è stato utilizzato un criterio diagnostico multifattoriale clinico-bioumorale con diversi parametri immunologici ed immunoistopatologico sulla biopsia, valutando la risposta terapeutica durante un follow-up longitudinale con uno score clinico-funzionale composito.
La DM può essere distinta dai segni clinici e dalle anomalie patologiche; si ritiene attualmente che essa sia il risultato di una vasculopatia.
Sulle biopsie muscolari sono stati caratterizzati con anticorpi monoclonali, le sottopopolazioni di linfociti T (CD4, CD8), i macrofagi, IgG, IgM, le frazioni C1q, C3, C4 del Complemento, gli antigeni di istocompatibilità MHC-I ed MHC-II.
Nelle PM l'immunità cellulo mediata era più pronunciata ed in alcuni casi, l'espressione di MHC-I ed MHC-II era osservabile anche sulla superficie ed all'interno di fibre muscolari.
I pazienti sono stati trattati, con steroidi e nei casi non responsivi, con aggiunta di azatioprina, ciclofosfamide, plasmaferesi, alte dosi di immunoglobuline endovena ed irradiazione “total body”.
Similar content being viewed by others
References
Appleyard S.T., Dubowitz V., Dunn M.J., Rose M.L.:Increased expression of HLA ABC class I antigens by muscle fibers in Duchenne muscular dystrophy, inflammatory myopathy, and other neuromuscular disorders. Lancet 1:361–363, 1985.
Arahata K., Engel A.G.:Monoclonal antibodies analysis of muclear cells in myopathies. I: quantitation of subsets according to diagnosis and sites of accumulation and demonstration and counts of muscle fibers invaded by T cell. Ann. Neurol. 16:193–208, 1984.
Bao S., King N.J.C., Dos Remedios C.G.:Elevated MHC class I and II antigens in cultured human embryonic myoblasts following stimulation with gamma-interferon. Immunol. Cell. Biol. 68:235–242, 1990.
Cherin P., Herson S., Wechsler B. et al.:Intravenous immunoglobulin for polymyositis and dermatomyositis. Lancet 336:116, 1990.
Dalakas M.C.:Treatment of polymyositis and dermatomyositis. Curr. Opin. Rheumatol. 1:444–9, 1989.
Dalakas M.C.:Polymyositis, Dermatomyositis and inclusion body myositis. N. Engl. J. Med. 325:1487–1498, 1991.
Dau P.C.:Plasmapheresis in idiopathic inflammatory myopathies: experience with 35 patients. Arch. Neurol. 38:544–52, 1981.
Emslie-Smith A.M., Arahata K., Engel A.G.:Major histocompatibility complex class I antigen expression, immunolocalization of interferon subtypes, and T cell-mediated cytotoxicity in myopathies. Hum. Pathol. 20:224–231, 1989.
Emslie-Smith A.M., Engle A.G.:Necrotizing myopathy with pipestem capillaries, microvascular deposition of the complement membrane attack complex (MAC), and minimal cellular infiltration. Neurology 41:936–939, 1991.
Engel A.G., Arahata K.:Mononuclear cells in myopathies: quantitation of functionally distinct subsets, recognition of antigen-specific cell-mediated cytotoxicity in some diseases and implications for the pathogenesis of the different inflammatory myopathies. Hum. Pathol. 17:704–721, 1986.
Engel A.G., Emslie-Smith A.M.:Inflammatory myopathies. Curr. Opin. Neurol. Neurosurg. 2:695–700, 1989.
Engel W.K., Lichter A.S., Galdi A.P.:Polymyositis: remarkable response to total body irradiation. Lancet 1:658, 1981.
Hohlfeld R., Engel A.G.:Induction of HLA-DR expression on human myoblasts with interferongamma. Am. J. Pathol. 136:503–508, 1990.
Hubbard W.N., Walport M.J., Halnnan K.E. et al.:Remission from polymyositis after total body irradiation. BMJ 284:1915–6, 1982.
Jan S., Beretta S., Maggio M. et al.:High dose intravenous human immunoglobulin in treatmentresistant polymyositis. Neurology 40: suppl. 1:120, 1990.
Karpati G., Pouliot Y., Carpenter S.:Expression of immunoreactive major histocompatibility complex products in human skeletal muscles. Ann. Neurol. 23:64–72, 1988.
Kelly J.J., Madoc-Jones H., Adelman L.S. et al.:Response to total body irradiation in dermatomyositis. Muscle & Nerve 11: 120–3, 1988.
Mantegazza R., Hughes S.M., Mitchell D. et al.:Modulation of MHC class II antigen expression in human myoblasts after treatment with interferon-gamma. Neurology 41:1128–1132, 1991.
McDouall R.M., Dunn M.J., Dubowitz V.:Expression of class I and class II MHC antigens in neuromuscular diseases. J. Neurol. Sci. 89:213–226, 1989.
Walton J.:The idiopathic inflammatory myopathies and their treatment. J. Neurol. Neurosurg. Psychiatry 54:285–7, 1991.
Zuk J.A., Fletcher A.:Skeletal muscle expression of class II histocompatibility (HLA-DR) in polymyositis and other muscle disorders with an inflammatory infiltrate. J. Clin. Pathol. 41:410–414, 1988.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Angelini, C., Menegazzo, E. & Fanin, M. Multifactorial study of inflammatory myopathies. Report of 29 cases. Ital J Neuro Sci 14, 69–76 (1993). https://doi.org/10.1007/BF02339045
Issue Date:
DOI: https://doi.org/10.1007/BF02339045