Summary
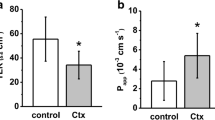
Protamine is a naturally occurring basic protein (pI; 9.7 to 12.0). We have recently reported that protamine dissolved in the mucosal bath (2 to 20 μm), induces about a twofold increase in transepithelial resistance inNecturus gallbladder within 10 min. Conductance decreased concomitantly with cation selectivity.
In this leaky epithelium, where >90% of an applied current passes between cells, an increment in resistance of this magnitude suggests a paracellular actiona priori. To confirm this, ionic conductance across the apical cell membrane was studied with microelectrodes. Protamine increased transepithelial resistance without changing apical cell membrane voltage or fractional membrane resistance. Variation in extracellular K concentration (6 to 50mm) caused changes in apical membrane voltage not different from control.
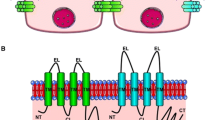
To determine if protamine-induced resistance changes were associated with structural alteration of tight junctions, gallbladders were fixedin situ at peak response and analyzed by freeze-fracture electron microscopy. According to a morphometrical analysis, the tight junctional intramembranous domain expands vertically due to incorporation of new strands (fibrils) into the main compact fibrillar meshwork.
Since morphologic changes are complete within 10 min, strands are probably recycled into and out of the tight junctional membrane domain possibly by the cytoskeleton either from cytoplasmic vesicles or from intramembranous precursors. Regulation of tight junctional permeability by protamine and other perturbations may constitute a common mechanism by which leaky epithelia regulate transport, and protamine, in concentrations employed in this study, seems reasonably specific for the tight junction.
Similar content being viewed by others
References
Alavi, N., Lianos, E.A., Palant, C.E., Bentzel, C.J. 1983. Induction of epithelial tight junctions by a light chain protein isolated from a patient with Fanconi's syndrome.Nephron 35:130–135
Bentzel, C.J., Hainau, B., Ho, S., Hui, S.W., Edelman, A., Anagnostopoulos, T., Benedetti, E.L. 1980. Cytoplasmic regulation of tight-junction permeability: Effect of plant cytokinins.Am. J. Physiol. 239:C75-C89
Boulpaep, E.L., Sackin, H. 1980. Electrical analysis of intraepithelial barriers.In: Current Topics in Membrane and Transport. F. Bronner, A. Kleinzeller, and E.L. Boulpaep, editors. pp. 169–197. Academic, New York
Chalcroft, J.P., Bullivant, S. 1970. An interpretation of liver cell membrane and junction structure based on observation of freeze fracture replicas of both sides of the fracture.J. Cell. Biol. 47:49–60
Claude, P. 1978. Morphological factors influencing transepithelial permeability: A model for the resistance of the zonula occludens.J. Membrane Biol. 39:219–232
Duffey, M.E., Hainau, B., Ho, S., Bentzel, C.J. 1981. Regulation of epithelial tight junction permeability by cyclic AMP.Nature (London) 294:451–453
Fromm, M., Palant, C.E., Bentzel, C.J., Hegel, U. 1985. Protamine reversibly decreases paracellular cation permeability inNecturus gallbladder.J. Membrane Biol. 87:141–150
Fromm, M., Weskamp, P., Hegel, U. 1980. Versatile piezoelectric driver for cell puncture.Pfluegers Arch. 384:69–73
Graf, J., Giebisch, G. 1979. Intracellular sodium activity and sodium transport inNecturus gallbladder epithelium.J. Membrane Biol. 47:327–355
Kachar, B., Reese, T.S. 1982. Evidence for the lipidic nature of tight junction strands.Nature (London) 296:464–466
Madara, J.L. 1983. Increases in guinea pig small intestinal transepithelial resistance induced by osmotic loads are accompanied by rapid alterations in absorptive-cell tight junction structure.J. Cell Biol. 97:125–136
Martinez-Palomo, A., Meza, I., Beaty, G., Cereijido, M. 1980. Experimental modulation of occluding junctions in a cultured transporting epithelium.J. Cell. Biol. 87:736–745
Meza, I., Ibarra, G., Sabanero, M., Martinez-Palomo, A., Cereijido, M. 1980. Occluding junctions and cytoskeletal components in a cultured transporting epithelium.J. Cell Biol. 87:746–754
Meza, I., Sabanero, M., Stefani, E., Cereijido, M. 1982. Occluding junctions in MDCK cells: Modulation of transepithelial permeability of transepithelial permeability by the cytoskeleton.J. Cell Biochem. 18:407–421
Moreno, J.H. 1975. Blockage of gallbladder tight junction cation-selective channels by 2,4,6-triaminopyrimidinium (TAP).J. Gen. Physiol. 66:97–115
Muller, J., Kachadorian, W.A., DiScala, V.A. 1980. Evidence that ADH-stimulated intramembrane particle aggregates are transferred from cytoplasmic to luminal membranes in toad bladder epithelial cells.J. Cell Biol. 85:83–95
Nagel, W., Garcia-Diaz, F.J., Essig, A. 1983. Contribution of junctional conductance to the cellular voltage-divider ratio in frog skins.Pfluegers Arch. 399:336–341
Palant, C.E., Duffey, M.E., Mookerjee, B.K., Bentzel, C.J. 1983. Ca2+ regulation of tight-junction permeability and structure inNecturus gallbladder.Am. J. Physiol. 245:C203-C212
Poler, S.M., Reuss, L. 1985. Protamine decreases apical membrane K+ and increases apical membrane Cl− conductance inNecturus gallbladder epithelium.J. Gen. Physiol. 86:42a
Reuss, L., Finn, A.L. 1975. Electrical properties of the cellular transepithelial pathway inNecturus gallbladder: I. Circuit analysis and steady-state effects of mucosal solution ionic substitutions.J. Membrane Biol. 25:115–139
Reuss, L., Grady, T.P. 1979. Triaminopyrimidinium (TAP+) blocks luminal membrane K conductance inNecturus gallbladder epithelium.J. Membrane Biol. 48:285–298
Suzuki, K., Frömter, E. 1977. The potential and resistance profile ofNecturus gallbladder cells.Pfluegers Arch. 371:109–117
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bentzel, C.J., Fromm, M., Palant, C.E. et al. Protamine alters structure and conductance ofNecturus gallbladder tight junctions without major electrical effects on the apical cell membrane. J. Membrain Biol. 95, 9–20 (1987). https://doi.org/10.1007/BF01869626
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01869626