Summary
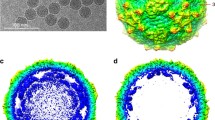
Polypeptide synthesis and morphogenesis of a group C rotavirus (AmC-1) adapted to a continuous swine testicular cell line was examined. SDS-PAGE analysis of35S methionine labeled infected cell lysates revealed 9 viral polypeptides (122, 98, 79, 78, 43, 41, 35, 24, and 20 kD). Viral polypeptide synthesis appeared to be maximal at 7–10 h post infection. Purified group C virus grown in the presence of trypsin was found to contain seven structural polypeptides (122, 98, 79, 53, 43, 41, and 30 kD) by protein blotting and five polypeptides (98, 79, 78, 43, and 41 kD) by immunoprecipitation with a hyperimmune rabbit antisera. Tunicamycin treatment, Concanavalin A binding, protein blotting, endo-H treatment and2,6H-mannose labeling suggested that group C rotavirus contains one structural glycoprotein (41 kD) with a corresponding precursor mol. wt. of 37 kD and one not previously identified non-structural glycoprotein (24 kD) with a corresponding precursor mol. wt. of ⩽20 kD. Electron microscopy of infected swine testicular cells revealed an assembly process for group C rotavirus similar to group A, with single-shelled particles budding through the rough endoplasmic reticulum with concomitant acquisition of a transient membrane.
Similar content being viewed by others
References
Au KS, Chan WK, Burns JW, Estes MK (1989) Receptor activity of rotavirus non-structural glycoproteins NS 28. J Virol 63: 4553–4562
Bellamy AR, Both GW (1990) Molecular biology of rotaviruses. Adv Virus Res 38: 1–34
Bremont M, Cohen J, McCrae MA (1988) Analysis of the structural polypeptides of a porcine group C rotavirus. J Virol 62: 2183–2185
Bremont M, Juste-Lesage P, Chabanne-Vautherot D, Charpilienne A, Cohen J (1992) Sequences of the four larger proteins of a porcine group C rotavirus and comparison with the equivalent group A rotavirus proteins. Virology 186: 684–692
Bridger JC, Pedley S, McCrae MA (1986) Group C rotaviruses in humans. J Clin Microbiol 23: 760–763
Caul EO, Ashley CR, Darville JM, Bridger JC (1990) Group C rotavirus associated with fatal enteritis in a family outbreak. J Med Virol 30: 201–205
Cooke SJ, Lambden PR, Caul EO, Clarke IN (1991) Molecular cloning, sequence and coding assignment of the major inner capsid protein gene of human group C rotavirus. Virology 184: 781–785
Ericson CL, Graham DY, Mason BB, Estes MK (1982) Identification, synthesis, and modifications of simian rotavirus SA 11 polypeptides in infected cells. J Virol 42: 825–839
Espejo RT, Puerto F, Soler C, Gonzalez N (1984) Characterization of a human pararotavirus. Infect Immun 44: 112–116
Estes MK, Graham DY, Mason BB (1981) Proteolytic enhancement of rotavirus infectivity: molecular mechanisms. J Virol 39: 879–888
Jiang BM, Saif LJ, Kang SY, Kim JH (1990) Biochemical characterization of the structural and nonstructural polypeptides of a porcine group C rotavirus. J Virol 64: 3171–3178
Matsumoto K, Hatano M, Kobayashi K, Hasegawa A, Yamazaki S, Nakata S, Chiba S, Kimura Y (1989) An outbreak of gastroenteritis associated with acute rotaviral infection in schoolchildren. J Infect Dis 160: 611–615
Maunula L, Svensson L, Bonsdorff C-H (1992) A family outbreak of gastroenteritis caused by group C rotavirus. Arch Virol 124: 269–278
Meyer JC, Bergmann CC, Bellamy AR (1989) Interaction of rotavirus cores with the nonstructural glycoprotein NS 28. Virology 171: 98–107
Poruchynsky MS, Tyndall C, Both GW, Sato F, Bellamy AR, Atkinson PH (1985) Deletions into an NH 2-terminal hydrophobic domain result in secretion of rotavirus VP 7, a resident endoplasmic reticulum membrane glycoprotein. J Cell Biol 101: 2199–2209
Qian Y, Jiang B, Saif LJ, Kang SY, Ishimaru Y, Yamashita Y, Oseto M, Green KY (1991) Sequence conservation of gene 8 between human and porcine group C rotaviruses and its relationship to the vp 7 gene of group A rotaviruses. Virology 182: 562–569
Saif LJ (1990) Nongroup A rotaviruses. In: Saif JL, Thiel KW (eds) Viral diarrheas of man and animals. CRC Press, Boca Raton, pp 73–95
Saif LJ, Terrett LA, Miller KL, Cross RF (1988) Serial propagation of porcine group C rotavirus (pararotavirus) in a continuous cell line and characterization of the passaged virus. J Clin Microbiol 26: 1277–1282
Stirzaker SC, Both GW (1989) The signal peptide of the rotavirus glycoprotein VP 7 is essential for its retention in the ER as an integral membrane protein. Cell 56: 741–747
Svensson L (1984) Identification of an outer capsid glycoprotein of human rotavirus by concanavalin A. J Gen Virol 65: 2183–2190
Svensson L (1992) Group C rotavirus requires sialic acid for erythrocyte and cell receptor binding. J Virol 66: 5582–5585
Svensson L, Sheshberadaran H, Vene S, Norrby E, Grandien M, Wadell G (1987) Serum antibody responses to individual viral polypeptides in human rotavirus infections. J Gen Virol 68: 643–651
Terrett LA, Saif LJ (1987) Serial propagation of porcine group C rotavirus (pararotavirus) in primary porcine kidney cell cultures. J Clin Microbiol 25: 1316–1319
Tsunemitsu H, Saif LJ, Jiang B, Shimizu M, Hiro M, Yamaguchi H, Ishiyama T, Hirai T (1991) Isolation, characterization, and serial propagation of a bovine group C rotavirus in a monkey kidney cell line (MA-104). J Clin Microbiol 29: 2609–2613
von Bonsdorff C-H, Svensson L (1988) Human serogroup C rotavirus in Finland. Scand J Infect Dis 20: 475–478
Welter M, Welter J, Chambers D, Svensson L (1991) Adaptation and serial passage of porcine group C rotavirus in ST cells, an established diploid swine testicular cell line. Arch Virol 120: 297–304
Whitefeld PL, Tyndall C, Stirzaker SC, Bellamy AR, Both GW (1987) Location of sequences within rotavirus SA 11 glycoprotein VP 7 which direct it to the endoplasmic reticulum. Mol Cell Biol 7: 2491–2497
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nilsson, M., von Bonsdorff, C.H. & Svensson, L. Biosynthesis and morphogenesis of group C rotavirus in swine testicular cells. Archives of Virology 133, 21–37 (1993). https://doi.org/10.1007/BF01309741
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01309741