Summary
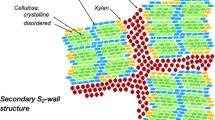
The study was devoted to the microstructure of the thick walled cells of the endocarp of prune (Prunus domestica L.), cherry (Prunus cerasus L.), walnut (Juglons regia L.). The tissue is formed of closely associated cells showing a homogeneous development characterized by an intense constructive activity of ordered walls with a typically twisted pattern (cholesteric-like). The arced layers are produced in tens, each corresponding to a 180° full rotation of the molecules (axis of rotation oriented radially) and their succession gives rise to a basic regular and monotonous periodicity. On the other hand, observation of the tissue revealed the large capacity of the helicoidal morphogenesis to adjust itself under the influence of two topological contingent constraints: (1) the spherical shape (and derivated shapes) of the cell and (2) the numerous pit canals which maintain the symplastic transport and produce a recess during the construction of the wall. Spherical shape (closed surfaces) and recess both introduce additional internal strains which are relieved by deviations of the molecular array in the basic pattern (moiré and knotty aspects). Special attention was given to the defects integrated in the spherical twist. The defects emerging in the angled stacks of microfibrils (disclinations, distortions) were a diagnostic feature of an actual liquid crystal behaviour under mechanical constraints. The abundance of such defects, of cusps and spiral motions strengthened the hypothesis that a transient fluid phase, later on consolidated and stiffened, operates during the cellulose ordering. The saddle-like figures developed in the complex polylobed situation of walnut were particularly demonstrative. The fractionation of the secondary wall yielded the glucidic matrix in the same ratio as cellulose. The bulk of this embedding matrix was composed of acidic xylans more or less tightly bound to the microfibrils. The coat of negatively charged polysaccharides visualized by the binding of cationic gold to wall strips might be expected to act as a surfactant generating an electrostatic repulsion between microfibrils. This could be a cooperative mechanism for the self-positioning (aligment in sheets and progressive rotation) of the composite.
Similar content being viewed by others
References
Abeysekera RM, Willison JHM (1988) Development of helicoidal texture in the prerelease mucilage of quince (Cydonia oblonga) seed epidermis. Can J Bot 66: 460–467
Aspinall GO (1970) Polysaccharides. Pergamon Press, Oxford
Aspinall GO (1980) Chemistry of polysaccharides. In: Preiss J (ed) The biochemistry of plants, vol 3. Academic Press, New York, pp 473–500
Atlan H (1974) On a formal definition of organization. J Theor Biol 45: 295–304
Bouligand Y (1972) Twisted fibrous arrangements in biological materials and cholesteric mesophases. Tissues Cell 4: 189–217
— (1978) Cholesteric order in biopolymer. In: Blumstein B (ed) Mesomorphic order in polymer and polymerisation in liquid crystalline media. American Chemical Society, Washington, DC, pp 261–297
Chalmers DJ, Van den Ende B (1977) The relation between seed and fruit development in the peach (Prunus persica L.). Ann Bot 41: 707–714
Coombe BG (1976) The development of fleshy fruit. Annu Rev Plant Physiol 27: 207–208
Das N, Das SC, Dutt AS, Roy A (1981) Lignin-xylan ester linkage in jute fiber (Corchorus capsularis). Carbohydr Res 94: 73–82
Dupont MS, Selvendran RR (1987) Hemicellulosic polymer from the cell walls of beeswing wheat bran: 1 Polymer solubilized by alkali. Carbohydr Res 163: 99–113
Folda T, Hoffmann H, Chanzy H, Smith P (1988) Liquid crystalline suspension of (tetrafluoroethylene) “whiskers”. Nature 6168: 55–56
Fry SC (1988) The growing plant cell wall: chemical and metabolic analysis. Longman, Essex
Gaill F, Hunt S (1988) Tubes. In: Westheide W, Hermans CO (eds) The ultrastructure of Polychaeta. G Fischer, Stuttgart, pp 61–70 (Microfauna Marina, vol 4)
Giraud-Guille MM (1986) Liquid crystalline phases of sonicated type I collagen. Biol Cell 67: 97–101
Gray DG (1983) Liquid crystalline cellulose. J Appl Polymer Sci 37: 179–192
Harris WF (1977) Disclinations. Sci Amer 237: 130–146
Herth W, Hausser I (1984) Chitin and cellulose fibrillogenesis in vivo and their experimental alteration. In: Dugger WM, Bartnicki-Garcia S (eds) Structure, function and biogenesis of plant cell walls. American Society of Plant Physiologists, Rockville, pp 89–120
- Jarvis MC (1991) Self-assembly of plant cell walls. Plant Cell Environ (in press)
Joseleau JP (1991) Chemical structure of xylan and their interaction with cellulose microfibrils. In: International symposium on xylans and xylanases, Wageningen, The Netherland, December 1991
Labecque M, Barabe D (1984) Développement du fruit dePrunus serotina (Rosacées). Can J Bot 62: 195–206
Lindberg B, Mosihuzzaman M, Nahar N, Abeysekera RM, Brown RG, Willison JH (1990) An unusual (4-O-methyl-D-glucurono)D-xylan isolated from the mucilage of seeds of the quince tree (Cydonia oblonga). Carbohydr Res 207: 307–310
Lloyd CW (1987) The plant cytoskeleton: the impact of fluorescence microscopy. Annu Rev Plant Physiol 38: 119–321
Marchessault RH, Liang CY (1962) The infrared spectra of cristalline polysaccharides. VIII. Xylans. J Polymer Sci 59: 357–378
Mazur GD, Regier JC, Kafatos FC (1982) Order and defects in the silkmoth chorion, a biological analogue of a cholesteric liquid crystal. In: King RC, Akai H (eds) Insect ultrastructure. Plenum Press, New York, pp 150–185
— — — (1989) Morphogenesis of silk chorion: sequential modification in an early helicoidal framework through expansion and densification. Tissue Cell 21: 227–242
Mora F, Ruel K., Comtat J, Joseleau JP (1986) Aspect of native and redeposited xylans at the surface of cellulose microfibrils. Holzforschung 40: 85–91
Neville AC (1986) The physics of helicoids. Phys Bull 37: 74–76
— (1988) A pipe-cleaner molecular model for morphogenesis of helicoidal plant cell wall based on hemicellulose complexity. J Theor Biol 131: 243–254
Nieduzinski IA, Marchessault RH (1972) Structure of D(1–4)-xylan hydrate. Biopolymers 11: 1335–1344
Northcote DH (1984) Control of cell wall assembly during differentiation. Giorn Bot Ital 118: 41–51
Parameswaran N, Sinner M (1979) Topological study of the wall of beech bark sclereid by enzymatic and acidic degradation. Protoplasma 101: 197–215
Quader HW, Herth W, Ryser U, Schnepf E (1987) Cytoskeletal elements in cotton seed hair development in vitro: their possible regulatory role of cell wall organization. Protoplasma 137: 56–62
Rees DA, Morris ER, Thom D, Madden JK (1982) Shapes and interactions of carbohydrates chains. In: Aspinall GO (ed) The polysaccharides. Academic Press, New York, pp 195–290
Reis D (1987) Cholesteric-like pattern in plant cell wall: different expressions. Mol Cryst Liq Cryst 153: 43–53
- Vian B, Chanzy H, Roland JC (1991) Liquid crystal-type assembly of native cellulose-glucuronoxylans extracted from plant cell walls. Biol Cell 73 (in press)
Roberts E, Seagull RW, Haigier CH, Brown RM (1982) Alteration of cellulose microfibril formation in eucaryotic cells. Calcofluor white interference with microfibril assembly and orientation inOocystis spiculata. Protoplasma 113: 1–9
Roland JC, Reis D, Vian B, Roy S (1989) The helicoidal plant cell wall as a performing cellulose-based composite. Biol Cell 67: 209–220
—, Satiat-Jeunemaitre B, Mosiniak M (1987) Morphogenesis of plant cell walls at the supramolecular level. Internal geometry and versatility of helicoidal expression. Protoplasma 140: 75–91
Rosell KG, Svensson S (1975) Studies of the distribution of 4-Omethyl-D-glucuronic acid residues in birch xylan. Carbohydr Res 42: 297–304
Skutelsky E, Roth J (1986) Cationic colloidal gold. A new probe for the detection of anonic cell surface sites by electron microscopy. J Histochem Cytochem 34: 693–696
Sterling C (1953) Developmental anatomy of the fruit ofPrunus domestica L. Bull Torrey Bot Club 80: 457–477
Vian B, Reis D (1991) Relationship of cellulose and other cell wall components: supramolecular organization. In: Haigier HC, Weimer PJ (eds) Biosynthesis and biodegradation of cellulose. Marcel Dekker, New York, pp 25–50
— —, Mosiniak M, Roland JC (1986) The glucuronoxylans and the helicoidal shift in cellulose microfibrils in linden wood: cytochemistry in muro and in isolated molecules. Protoplasma 131: 185–189
Werbowyj R, Gray DG (1984) Optical properties of hydroxylpropylcellulose liquid crystals. Cholesteric pitch and polymer concentration. Macromolecules 17: 1512–1520
Willison JHM, Abeysekera RM (1988) A liquid crystal containing cellulose in living plant tissue. J Polymer Sci 26: 71–75
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reis, D., Roland, J.C., Mosiniak, M. et al. The sustained and warped helicoidal pattern of a xylan-cellulose composite: the stony endocarp model. Protoplasma 166, 21–34 (1992). https://doi.org/10.1007/BF01320139
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01320139