Abstract
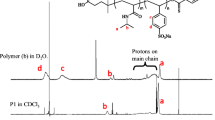
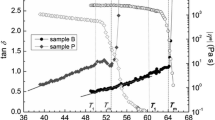
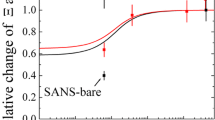
The reaction of the nonionic low molecular mass surfactant 3, 6, 9, 12, 15, 18, 21, 24-octaoxaoctatriacontane-1-ol (CH3(CH2)13(OCH2CH2)8OH, C14E8) with acryloyl chloride yielded the monomeric amphiphile II (CH3(CH2)13(OCH2CH2)8O2C-(CH:CH2) with the polymerizable group located at the hydrophilic end of the molecule. Using radical polymerization, the polymeric surfactant III is obtained. These three surfactant water systems exhibit lyotropic liquid crystalline phases. The binary phase diagrams are compared with each other. In changing from the monomer-water to the polymer-water system a stabilization of the lyotropic mesophases is observed with amphiphiles which are connected via their hydrophilic ends; it is known for the surfactants to be connected via their hydrophobic ends. The appearance of the inverse mesophases, as expected from the molecular geometry of the polymer, is not observed.
Similar content being viewed by others
References
Hall DG, Tiddy GJT (1981) (ed) Lucassen-Reynders EH, Surfactant Science Series 11, Anionic Surfactants, Marcel Dekker, New York and Basel, p 55–108
Tiddy GJT (1980) Physics Reports 57:1
Tiddy GJT, Walsh MF (1983) Studies in Physical and Theoretical Chemistry 26:151
Finkelmann H, Lühmann B, Rehage G (1982) Coll & Polym Sci 260:56
Lühmann B, Finkelmann H, Rehage G (1985) Makromol Chem 186:1059
Finkelmann H, Schafheutle MA (1986) Coll & Polym Sci 264:786
Still WC, Kahn M, Mitra A (1978) J Org Chem 43(14):2923
Mitchelll J, Tiddy GJT et al (1983) J Chem Soc Faraday Trans I:975
Lühmann B, Dissertation Clausthal (1985)
Tiddy GJT (1980) Physics Reports 57(1):28–29 (and cited literature)
Kjellander R, Florin E (1981) J Chem Soc Faraday Trans I 77:2053
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jahns, E., Finkelmann, H. Lyotropic liquid crystalline phase behavior of a polymeric amphiphile polymerized via their hydrophilic ends. Colloid & Polymer Sci 265, 304–311 (1987). https://doi.org/10.1007/BF01417929
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01417929