Abstract
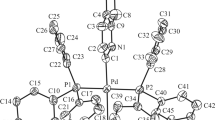
Anx-ray structural study of [QndH+]2[MoO 2−4 ]H2O (1) and [QndH+]Cl−H2O (2), where Qnd = quinidine, was conducted. The structures have an ionic structure, and the ions are joined by hydrogen bonds into chains (1) and layers (2). Quinidinium cations protonated at the quinuclidine N atom have a conformation with agauche position of the N atom and OH group (as in neutral quinidine) corresponding to the local, and not the global, minimum of the calculated conformational energy.
Similar content being viewed by others
Literature cited
K. Van Dyke, C. Szustkiewicz, C. H. Lantz, and L. H. Saxe,Biochem. Pharmacol.,18, No. 1, 147 (1969).
C. C. Cheng,J. Pharm. Sci.,60, No. 4, 1596 (1971).
B. J. Oleksyn and L. J. Lebioda,Polish J. Chem.,54, No. 4, 755 (1980).
S. Kashino and M. Haisa,Acta Crystallogr.,C39, No. 1, 310 (1983).
H. Ohbo, H. Okazaki, K. Miyoshi, and H. Yoneda,Bull. Chem. Soc. Jpn.,56, No. 7, 1982 (1983).
O. L. Carter, A. T. McPhail, and G. A.Sim, J. Chem. Soc. (A), No. 3, 365 (1967).
R. Doherty, W. R. Benson, M. Maienthal, and J. McD. Stewart,J. Pharm. Sci.,67, No. 5, 1698 (1978).
I. L. Karle and J. Karle,Proc. Nat. Acad. Sci. U.S.A.,78, 5938 (1981).
A. G. Orpen, L. Brammer, F. H. Allen, O. Kennard, D. G. Watson, and R. Taylor,J. Chem. Soc. Dalton Trans., No. 12, S1 (1989).
W. Hamilton,Acta Crystallogr.,18, No. 2, 502 (1965).
Additional information
A. N. Nesmeyanov Institute of Organoelemental Compounds, Russian Academy of Sciences, 117813 Moscow. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 5, pp. 1073–1078, May, 1992.
Rights and permissions
About this article
Cite this article
Shamuratov, É.B., Batsanov, A.S., Struchkov, Y.T. et al. Crystal structures of bis(quinidinium) molybdate hydrate and quinidinium chloride hydrate: The question of the stable conformation of quinidine. Russ Chem Bull 41, 841–845 (1992). https://doi.org/10.1007/BF00864530
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00864530