Abstract
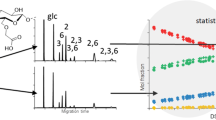
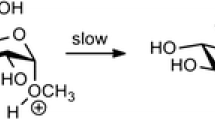
Monomer compositional data of a series of hydroxyethyl celluloses (HECs) with molar substitutions (MS) ranging from 0.2 to 2.4 were used to analyse the substituents in the samples. The data reveal that the reactivity of the pendant hydroxyl groups in the substituents decreases in progressed states of derivatization, and that the reactivity of the 6-O-positions is only large compared to the 2-O-positions in non-derivatized glucosyl residues. These two processes are not taken into account in any of the statistical models for the description of the substituent distribution in HECs, which shows that the assumption that the relative reaction constants of the various hydroxyl groups in HECs remain constant throughout the whole reaction is false. The occurrence of maxima in the mole fractions of the monomers was examined as a function of the MS of the samples by principal component analysis of the monomer compositional data. The results show that in the beginning phase of the derivatization mainly monosubstituted monomers are formed and that chain propagation of these substituents takes place, whereas in the progressed states of conversion mainly di- and trisubstituted moieties are formed. The changes in the reactivity of the various hydroxyl groups during the conversion of cellulose to HECs can be described by a model wherein the interactions of both NaOH and the diluents with the cellulosics are taken into account.
Similar content being viewed by others
References
Arisz, P. W. and Boon, J. J. (1993)Carbohydrates in the Netherlands,9, 38–42.
Arisz, P. W., Lomax, J. A. and Boon, J. J. (1993)Carbohydr. Res.,243, 99–114.
Dönges, R. (1990)Br. Polym. J.,23, 315–326.
Hodges, K. L., Rester, W. E., Wiederrich, D. L. and Grover, J. A. (1979)Anal. Chem.,51, 2172–2176.
Just, E. K. and Majewicz, T. G. (1985) InEncyclopedia of Polymer Science and Engineering, 2nd edn, vol. 3 (J. I. Kroschwitz, ed.) New York: Wiley, pp. 226–269.
Klug, E. D. (1966) U.S. Pat. 3, 278, 521 (Oct. 11, 1966 to Hercules Inc.)
Lindberg, B., Lindquist, U. and Stenberg, O. (1987)Carbohydr. Res.,5, 207–214.
Lorand, E. J. and Georgi, E. A. (1937)J. Am. Chem. Soc.,59, 1166–1170.
Ramnäs, O. and Samuelson, O. (1973)Svensk Papperstendning,15, 569–571.
Reuben, J. (1984)Macromolecules,17, 156–161.
Reuben, J. and Casti, T. E. (1987)Carbohydr. Res.,163, 91–98.
Spurlin, H. M. (1939)J. Am. Soc.,61, 2222–2227.
Spurlin, H. M. (1954) InCellulose and Cellulose Derivatives, Part II, (E. Ott, H. M. Spurlin and M. W. Graffin, eds.) New York: Interscience, p. 673–712.
Stratta, J. J. (1963)Tappi J.,46, 717–722.
Windig, W., Kistemaker, P. G. and Haverkamp, J. (1981/1982).J. Anal. Appl. Pyrolysis,3, 199–212.
Yokota, H. (1985)J. Appl. Polym. Sci.,30, 263–277.
Yokota, H. (1986a)J. Appl. Polym. Sci.,32, 3423–3433.
Yokota, H. (1986b)Cellulose Chem. Technol.,20, 315–325.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arisz, P.W., Thai, H.T.T., Boon, J.J. et al. Changes in substituent distribution patterns during the conversion of cellulose toO-(2-hydroxyethyl) celluloses. Cellulose 3, 45–61 (1996). https://doi.org/10.1007/BF02228790
Issue Date:
DOI: https://doi.org/10.1007/BF02228790