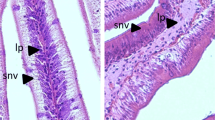
Abstract
Arctic char (Salvelinus alpinus L.) were fed over a three-week period with a commercial diet or one of seven casein-based diets. The latter were either lipid-free or supplemented with 50–200 g linseed oil kg−1 diet by dry weight, 160 g linseed oil and 40 g 14:0 or 160 g linseed oil and 40 g 16:0. Three fish having filled guts were sampled from each dietary group and analysed for ultrastructural changes in the pyloric caecum.
Increasing the dietary lipid level increased the accumulation of lipid droplets in columnar absorptive enterocytes from about 9% of epithelial volume in fish fed a diet of 50 g linseed oil, to almost 61% in fish fed a diet of 200 g linseed oil. Replacing linseed oil in the diet with 14:0 (160 g linseed oil + 40 g 14:0 kg−1 diet) appeared to produce a smaller lipid loading (roughly 53%) but the difference was not statistically significant. However, replacing 40 g linseed oil with 40 g of 16:0 in the diet decreased lipid loading significantly to just under 10%. Epithelial damage to enterocytes was assessed using a ranking scale based on ultrastructural signs of cell and organelle swelling and degeneration. The extent of damage closely followed the level of lipid loading, being lowest in fish fed the lipid-free or low-lipid (damage index 0.07–0.13) diets, and highest in char maintained on a diet containing 200 g linseed oil (index 1.41). Replacing linseed oil with 14:0 (160 g linseed oil + 40 g 14:0) appeared to reduce the damage index to 0.77 but this was not significant. However, a significant reduction of the damage index to 0.27 was observed when linseed oil was replaced by 16:0.
We conclude that higher dietary linseed oil promotes lipid droplet accumulation in enterocytes. The droplets are probably related to the amount of polyunsaturated fatty acids in the lipid. Intracellular droplet formation and cellular damage are both reduced by adding saturated fatty acids to the diet. This could be related to disruptions in the lipoprotein assembly rate. The cellular damages observed with high lipid diets are likely to be pathological and may lead to intestinal malfunction and represent a major infection route for pathogenic bacteria.
Similar content being viewed by others
References
Bergot, P. and Fléchon, J.E. 1970a. Forme et voie d'absorption intestinale des acides gras chez la truitte arc-en-ciel (Salmo gairdneriRich.). I. Lipides en particules. Ann. Biol. Anim. Biochim. Biophys. 10: 459-472.
Bergot, P. and Fléchon, J.E. 1970b. Forme et voie d'absorption intestinale des acides gras à chaine longue chez la truite arc-en-ciel (Salmo gairdneriRich.). II. Lipides ‘étalés’. Ann. Biol. Anim. Biochim. Biophys. 10: 473-480.
Chapman, M.J. 1980. Animal lipoproteins: chemistry, structure, and comparative aspects. J. Lipid Res. 21: 789-853.
Christie, W.W. 1982. The preparation of derivatives of lipid. In: Lipid Analysis, Isolation, Separation, Identification and Structural Analysis of Lipids. 2nd ed., pp. 51-62. Edited by W.W. Christie. Pergamon Press, Oxford.
Degrace, P., Caselli, C., Bernard, A. and Carlier, H. 1996a. Mesenteric lymph absorption of eicosapentaenoic acid in rats. Comp. Biochem. Physiol. 113A: 279-285.
Degrace, P., Caselli, C., Rayo, J. M. and Bernard, A. 1996b. Intestinal lymph absorption of butter, corn oil, cod liver oil, menhaden oil, and eicosapentaenoic and docosahexaenoic acid ethyl esters in rats. Lipids 31: 405-414.
Deplano, M., Diez, J. P., Connes, R., Kentouri-Divanach, M. and Cavalier, F. 1991. Appearance of lipid-absorption capacities in larvae of the sea bass, Dicentrarchus labraxduring transition to the exotrophic phase. Mar. Biol. 108: 361-371.
Folch, J., Lees, M. and Sloane-Stanley, G.H. 1957. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226: 497-509.
Fontagné, S., Geurden, I., Escaffre, A.-M. and Bergot, P. 1998. Histological changes induced by dietary phospholipids in intestine and liver of common carp (Cyprinus carpioL.) larvae. Aquaculture 161: 213-223.
Gundersen, H.J.G. and Jensen, E.B. 1987. The efficiency of systematic sampling and its prediction. J. Microsc. 147: 229-263.
Howard, C.V. and Reed, M.G. 1998. Unbiased Stereology. Three-Dimensional Measurement in Microscopy. Bios Scientific Publishers, Oxford.
Iijima, N., Gotou, T. and Kayama, M. 1995. Isolation and characterisation of serum lipoproteins in red sea bream. Fish. Sci. 61: 297-303.
Iwai, T. 1969. Fine structure of gut epithelial cells of larval and juvenile carp during absorption of fat and protein. Arch. Hist. Jap. 30: 183-199.
Kjørsvik, E., van der Meeren, T., Kryvi, H., Arnfinnson, J. and Kvenseth, P.G. 1991. Early development of the digestive tract of cod larvae, Gadus morhuaL., during start feeding and starvation. J. Fish Biol. 38: 1-15.
Loewe, H. and Eckmann, R. 1988. The ontogeny of the alimentary tract of coreogonid larvae: normal development. J. Fish Biol. 33: 841-850.
Mayhew, T.M. 1991. The new stereological methods for interpreting functional morphology from slices of cells and organs. Exp. Physiol. 76: 639-665.
Mayhew, T.M. 1992. A review of recent advances in stereology for quantifying neural structure. J. Neurocytol. 21: 313-328.
Mayhew, T.M., Myklebust, R., Whybrow, A. and Jenkins, R. 1999. Epithelial integrity, cell death and cell loss in mammalian small intestine. Histol. Histopathol. 14: 257-267.
McDowell, E.M. and Trump, B.R. 1976. Histological fixatives suitable for diagnostic light and electron microscopy. Arch. Pathol. Lab. Med. 100: 405-414.
Noaillac-Depeyre, J. and Gas, N. 1974. Fat absorption by the enterocytes of the carp (Cyprinus carpioL.). Cell Tiss. Res. 155: 353-365.
Noaillac-Depeyre, J. and Gas, N. 1976. Electron microscopic study on gut epithelium of the tench (Tinca tincaL.) with respect to its absorptive functions. Tiss. Cell 8: 511-530.
Noaillac-Depeyre, J. and Gas, N. 1979. Structure and function of the intestinal epithelial cells in the perch (Perca fluviatilisL.). Anat. Rec. 195: 621-640.
Olsen, R.E. and Ringø, E. 1997. Lipid digestibility in fish: A review. Recent Res. Devl. Lipid Res. 1: 199-265.
Olsen, R.E., Myklebust, R., Kaino, T. and Ringø, E. 1999. Lipid digestibility and ultrastructural changes in the enterocytes of Arctic char (Salvelinus alpinusL.) fed linseed oil and soybean lecithin. Fish Physiol. Biochem. 21: 35-44.
Sakr, S.W., Attia, N., Haourigui, M., Paul, J.L., Soni, T., Vacher, D. and Girard-Globa, A. 1997. Fatty acid composition of an oral load affects chylomicron size in human subjects. Br. J. Nutr. 77: 19-31.
Sarasquete, M.C., Polo, A. and YÚfera, M. 1995. Histology and histochemistry of the development of the digestive system of larval gilthead seabream, Sparus aurataL. Aquaculture 130: 79-92.
Sire, M.F. and Vernier, J.M. 1981. Etude ultrastructurale de la synthèse de chylomicrons au cours de l'absorption intestinale des lipides chez la truite. Influence de la nature des acides gras ingérés. Biol. Cell 40: 47-62.
Sire, M.F., Lutton, C. and Vernier, J.M. 1981. New views on intestinal absorption of lipids in teleostan fishes: an ultrastructural and biochemical study in the rainbow trout. J. Lipid Res. 22: 81-94.
Sokal, R.R. and Rohlf, F.J. 1981. Biometry. W.H. Freeman and Co., San Francisco.
Stroband, H.W.J. and Dabrowski, K.R. 1979. Morphological and physiological aspects of the digestive system and feeding in fresh-water fish larvae. In: Nutrition des Poissons. pp. 335-374. Edited by M. Fontaine. Centre National de la Recherche Scientifique, Paris. (Act. Colloque CNERNA).
Stroband, H.W.J. and Debets, F.M.H. 1978. The ultrastructure and renewal of the intestinal epithelium of the juvenile grasscarp, Ctenopharyngodon idella(Val.). Cell Tiss. Res. 187: 181-200.
Van Greevenbroek, M.M.J., Voorhout, W.F., Erkelens, D.W., van Meer, G. and de Bruin, T.W.A. 1995. Palmitic acid and linoleic acid metabolism in Caco-2 cells: different triglyceride synthesis and lipoprotein secretion. J. Lipid Res. 36: 13-24.
Watanabe, Y. and Sawada, N. 1985. Larval development of digestive organs and intestinal absorptive functions in the freshwater goby, Chaenogobius annularis.Bull. Tokoku Reg. Fish. Res. Lab. 47: 1-10.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Olsen, R., Myklebust, R., Ringø, E. et al. The influences of dietary linseed oil and saturated fatty acids on caecal enterocytes in Arctic char (Salvelinus alpinus L.): a quantitative ultrastructural study. Fish Physiology and Biochemistry 22, 207–216 (2000). https://doi.org/10.1023/A:1007879127182
Issue Date:
DOI: https://doi.org/10.1023/A:1007879127182