Abstract
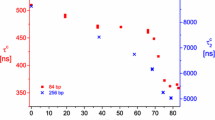
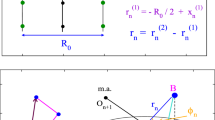
We present here recent results on the structure of superhelical DNA and its changes with salt concentration between 0.01 and 1.5 M NaCl. Scattering curves of two different superhelical DNAs were determined by static light scattering. The measured radii of gyration do not change significantly with salt concentration. Small-angle neutron scattering, together with calculations from a Monte Carlo model, allows to determine the superhelix diameter. Measured and simulated scattering curves agreed almost quantitatively. Experimentally we find that the diameter decreases from 16.0±0.9 nm at 10 mM to 9.0±0.7 nm at 100 mM NaCl. The superhelix diameter from the simulated conformations decreased from 18.0±1.5 nm at 10 mM to 9.4±1.5 nm at 100 mM NaCl. At higher salt concentrations up to 1.5 M NaCl, the diameter stays constant at 9 nm.
Similar content being viewed by others
References
Adrian, M., B. ten Heggeler-Bordier, W. Wahli, A.Z. Stasiak, A. Stasiak & J. Dubochet, 1990. Direct visualization of supercoiled DNA molecules in solution. EMBO J. 9: 4551–4554.
Bednar, J., P. Furrer, A. Stasiak, J. Dubochet, E.H. Egelman & A.D. Bates, 1994. The twist, writhe and overall shape of superhelical DNA change during counterion-induced transition from a loosely to a tightly interwound superhelix. Possible implications for DNA structure in vivo. J. Mol. Biol. 235: 825–847.
Boles, T.C., J.H. White & N.R. Cozzarelli, 1990. Structure of plectonemically supercoiled DNA. J. Mol. Biol. 213: 931–951.
Cantor, C.R. & P.R. Schimmel, 1980. Biophysical Chemistry. Part II: Techniques for the Study of Biological Structure and Function. Freeman, New York.
Chirico, G. & J. Langowski, 1996. Brownian dynamics simulations of supercoiled DNA with bent sequences. Biophys J. 71: 955–971.
Gebe, J.A., J.J. Delrow, P.J. Heath, B.S. Fujimoto, D.W. Stewart & J.M. Schurr, 1996. Effects of Na+ and Mg2+ on the structures of supercoiled DNAs: comparison of simulations with experiments. J. Mol. Biol. 262: 105–128.
Hammermann, M., N. Brun, K.V. Klenin, R. May, K. Tóth & J. Langowski, 1998. Salt-dependent DNA superhelix diameter studied by small angle neutron scattering measurements and Monte Carlo simulations. Biophys. J. 75: 3057–3063.
Hammermann, M., C. Steinmaier, H. Merlitz, U. Kapp, W. Waldeck, G. Chirico & J. Langowski, 1997. Salt effects on the structure and internal dynamics of superhelical DNAs studied by light scattering and Brownian dynamics. Biophys. J. 73: 2674–2687.
Klenin, K., H. Merlitz & J. Langowski, 1998. A Brownian dynamics program for the simulation of linear and circular DNA and other wormlike chain polyelectrolytes. Biophys. J. 74: 780–788.
Klenin, K.V., M.D. Frank-Kamenetskii & J. Langowski, 1995. Modulation of intramolecular interactions in superhelical DNA by curved sequences: A Monte Carlo simulation study. Biophys. J. 68: 81–88.
Rippe, K., N. Mucke & J. Langowski, 1997. Superhelix dimensions of a 1868 base pair plasmid determined by scanning force microscopy in air and in aqueous solution. Nucleic Acids Res. 25: 1736–1744.
Rybenkov, V.V., A.V. Vologodskii & N.R. Cozzarelli, 1997a. The effect of ionic conditions on the conformations of supercoiled DNA.1. Sedimentation analysis. J. Mol. Biol. 267: 299–311.
Rybenkov, V.V., A.V. Vologodskii & N.R. Cozzarelli, 1997b. The effect of ionic conditions on the conformations of supercoiled DNA. 2. Equilibrium catenation. J. Mol. Biol. 267: 312–323.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Langowski, J., Hammermann, M., Klenin, K. et al. Superhelical DNA studied by solution scattering and computer models. Genetica 106, 49–55 (1999). https://doi.org/10.1023/A:1003720610089
Issue Date:
DOI: https://doi.org/10.1023/A:1003720610089