Abstract
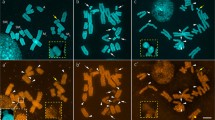
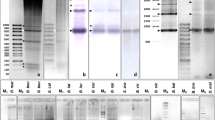
The DNA composition and the in situ hybridization of satellite fractions were analysed in the New World camelids llama, alpaca, guanaco and vicuña. In the four camelid forms, it was possible to identify a similar main band DNA and five satellite fractions (I–V) with G+C base contents ranging from 32% to 66%. Satellites II–V from llama were in situ reannealed on chromosomes from the four camelid forms. The results obtained were: (a) the four satellites hybridized with regions of C-banding (centromeric regions of all chromosomes and short arms of some autosomes); (b) in general, homologous hybridizations (llama DNA versus llama chromosomes) were more efficient than heterologous reassociations; there were however three exceptions to this rule (vicuña and alpaca satellite fraction II, chromosome group B; vicuña fraction V, chromosome groups A and B); (c) X chromosomes from the four camelids had satellites III–V but lacked satellite II, (d) no satellite fraction was detected on chromosome Y. The analysis of the in situ hybridization patterns allowed to conclude that most or all C-banded chromosome regions comprise several satellite DNA fractions. It is, moreover, proposed that there is an ample interspecies variation in the number of chromosomes that cross-react with a given satellite fraction. Our data give further support to the close genomic kinship of New World camelids.
Similar content being viewed by others
References
Balmain, A. & Birnie, G. D., 1979. Nick translation of mammalian DNA. Biochem. Biophys. Acta 561: 155–166.
Bianchi, N. O., Bianchi, M. S., Cleaver, J. E. & Wolff, S., 1985. The pattern of restriction enzyme-induced banding in the chromosomes of chimpanzee, gorilla and orangutan and its evolutionary significance. J. mol. Evol. 22: 323–333.
Bianchi, N. O., Larramendy, M. L., Bianchi, M. S. & Cortés, L., 1986. Karyological conservatism in South American camelids. Experientia 42: 622–624.
Bunch, T. D., Foote, W. C. & Maciulis, A., 1985. Chromosome banding pattern homologies and NORs for the bactrian camel, guanaco and llama. J. Heredity 76: 115–118.
Cordeiro-Stone, M. & Lee, C. S., 1976. Studies on the satellite DNAs of Drosophila nasutoides: their buoyant densities, melting temperatures, reassociation rates and localizations in polytene chromosomes. J. mol. Biol. 104: 1–24.
Franklin, W. L., 1982. Biology, ecology and relationship to man of the South American camelids. In: M. A. Mares & H. H. Genoways (eds.) Mammalian biology in South America, Vol. 6. The Pymatuniang Symposia in Ecology, Pittsburgh: 457–489.
Gosden, J. R., Mitchell, A. R., Seuánez, H. N. & Gosden, CH. M., 1977. The distribution of sequences complementary to human satellite DNAs I, II and IV in the chromosomes of chimpanzee (Pan troglodytes), gorilla (Gorilla gorilla) and orang utan (Pongo pygmaeus). Chromosoma (Berl.) 63: 253–271.
Harper, M. E. & Saunders, G., 1981. Localization of single copy DNA sequences on G-banded human chromosomes by in situ hybridization. Chromosoma (Berl.) 83: 431–439.
Kopecka, H., Macaya, G., Cortadas, J., Thiery, J. P. & Bernardi, G., 1978. Restriction enzyme analysis of satellite DNA components from the bovine genome. Eur. J. Biochem. 84: 189–195.
Kurnit, D. M., Shafit, B. R. & Maio, J. J., 1973. Multiple satellite deoxyribonucleic acids in the calf and their relation to the sex chromosomes. J. mol. Biol. 81: 273–284.
Kurnit, D. M., Brown, F. L. & Maio, J. J., 1978. Mammalian repetitive DNA sequences in a stable Robertsonian system. Characterization, in situ hybridization, and cross-species hybridizations of repetitive DNAs in calf, sheep and goat chromosomes. Cytogenet. Cell Genet. 21: 145–167.
Larramendy, M., Vidal-Rioja, L., Bianchi, M. & Bianchi, N. O., 1984. Camélidos Sudamericanos: Estudios Genéticos. Boletin de Lima 35: 92–96.
Macaya, G., Cortadas, J. & Bernardi, G., 1978. An analysis of the bovine genome by density-gradient centrifugation. Preparation of the dG+dC-rich DNA components. Eur. J. Biochem. 84: 179–188.
Mandel, M. & Marmur, J., 1968. Use of ultraviolet absorbance-temperature profiles for determining the guanine plus cytosine content of DNA. Methods in Enzymology 12, B: 195–206.
Manuelidis, L., 1977. A simplified method for preparation of mouse satellite DNA. Analyt. Biochem. 78: 561–568.
Novoa, C. & Wheeler, J. C., 1984. Lama and Alpaca. In: IanMason (ed.) Evolution of the domesticated animals. Longman, London and New York: 116–128.
Pivec, L., Stokrova, J. & Sormova, Z., 1972. Plurimodal heterogeneity of base composition of calf thymus DNA. Biochim. biophys. Acta 272: 179–190.
Romero, E. C., 1927. Llamas, alpacas, vicuñas y guanacos. Tesis Doctoral, Fac. Agr. C. Vet., Buenos Aires.
Taylor, K. M., Hungerford, D. A., Snyder, R. L. & Ulmer, F. A., 1968. Uniformity of karyotypes in the Camelidae. Cytogenetics 7: 8–15.
Vidal-Rioja, L., Semorile, L. & Bianchi, N. O., 1979. DNA of Akodon (Rodentia, Cricetidae). I. Biophysical characterization. Biochem. Genet. 17: 111–131.
Vidal-Rioja, L., Bianchi, N. O., Catalá, A. & Semorile, L., 1982. DNA of Akodon (Rodentia, Cricetidae). II. Molecular hybridization of repetitive DNA sequences. Can. J. Genet. Cytol. 24: 601–609.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vidal-Rioja, L., Semorile, L., Bianchi, N.O. et al. DNA composition in South American camelids I. Characterization and in situ hybridization of satellite DNA fractions. Genetica 72, 137–146 (1987). https://doi.org/10.1007/BF00123172
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00123172