Abstract
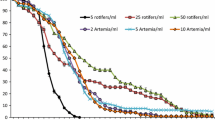
The contribution of ciliates as a food source to survival of first-feeding surgeonfish larvae, Paracanthurus hepatus, was examined in rearing experiments. The larvae were exposed to eight treatments; i.e. a tintinnid, Amphorellopsis acuta (1.0 × 104, 5.1 × 103 and 2.2 × 103 cells l−1) and a naked ciliate, Euplotes sp. (1.3 × 104, 8.0 × 103 and 5.0 × 103 cells l−1), plus two controls without ciliates. Highest survival of the larvae over the first 4–8 days was observed in the highest density of A. acuta. Rearing experiments also showed that the survivals of larvae fed with A. acuta were higher than those fed with Euplotes sp. Gut content analyses revealed loricae of A. acuta in the larvae. Although Euplotes sp. (lacking loricae) was never recognized in those larval guts, feeding on Euplotes sp. by larvae was confirmed using the ciliate labeled with fluorescent microspheres, implying that the feeding on naked ciliates by fish larvae has been overlooked. The results strongly suggested that both tintinnid and naked ciliates play important roles as alternative food sources to copepod nauplii by enhancing the survivability of fish larvae, especially those with a smaller mouth.
Similar content being viewed by others
References
Arthur, D. K., 1977. Distribution, size, and abundance of microcopepods in the California Current system and their possible influence on survival of marine teleost larvae. Fish. Bull. U.S.A. 75: 601–611.
Bagarinao, T., 1986. Yolk resorption, onset of feeding and survival potential of larvae of three tropical marine fish species reared in the hatchery. Mar. Biol. 91: 449–459.
Bailey, K. M. &; C. L. Stehr, 1986. Laboratory studies on the early life history of the walleye pollock, Theragra chalcogramma (Pallas). J. exp. mar. Biol. Ecol. 99: 233–246.
Dolan, J. R. &; D. W. Coats, 1991. A study of feeding in predacious ciliates using prey ciliates labeled with fluorescent microspheres. J. Plankton Res. 13: 609–627.
Funakoshi, S., 1984. Food and survival of the early postlarvae of the Japanese anchovy, Engraulis japonica (Houttuyn)-II. Distribution of anchovy larvae as related to occurrence of the maximum layers of chlorophyll-a concentration and copepod nauplius densities. Bull. Japan. Soc. Fish. Oceanogr. 45: 21–30 (in Japanese with English abstract).
Govoni, J. J. &; A. J. Chester, 1990. Diet composition of larval Leiostomus xanthurus in and about the Mississippi River plume. J. Plankton Res. 12: 819–830.
Hunter, J. R., 1977. Behavior and survival of northern anchovy Engraulis mordax larvae. Calif. Coop. Oceanic Fish. Invest. Rep. 19: 138–146.
Hunter, J. R., 1981. Feeding ecology and predation of marine fish larvae. In Lasker, R. (ed.), Marine Fish Larvae. University of Washington Press, Seattle and London: 33–77.
Hunter, J. R. &; C. A. Kimbrell, 1980. Early life history of Pacific mackerel, Scomber japonicus. Fish. Bull. U.S.A. 78: 89–101.
Jenkins, G. P., 1987. Comparative diets, prey selection, and predatory impact of co-occurring larvae of two flounder species. J. exp. mar. Biol. Ecol. 110: 147–170.
Kamiyama, T., 1994. The impact of grazing by microzooplankton in northern Hiroshima Bay, the Seto Inland Sea, Japan. Mar. Biol. 119: 77–88.
Kentouri, M. &; P. Divanach, 1983. Contribution à la connaissance du comportement et de la biologie des larves de marbré Lithognathus mormyrus (sparides) en élevage. Ann. Zootech. 32: 135–152.
Kleppel, G. S., 1993. On the diets of calanoid copepods. Mar. Ecol. Prog. Ser. 99: 183–195.
Kohno, H., A. Ohno &; Y. Taki, 1994. Why is grouper larval rearing difficult?: a comparison of the biological natures of early larvae of four tropical marine fish species. In Chou L. M., A. D. Munro, T. J. Lam, T. W. Chen, L. K. K. Cheong, J. K. Ding, K. K. Hooi, H. W. Khoo, V. P. E. Phang, K. F. Shin &; C. H. Tan (eds), The Third Asian Fisheries Forum. Asian Fisheries Society, Manila, Philippines: 450–453.
Kohno, H., S. Hara, M. Duray &; A. Gallego, 1988. Transition from endogenous to exogenous nutrition sources in larval rabbitfish Siganus guttatus. Nippon Suisan Gakkaishi. 54: 1083–1091.
Kohno, H., S. Diani, P. Sunyoto, B. Slamet &; P. T. Imanto, 1990. Early developmental events associated with changeover of nutrient sources in the grouper, Epinephelus fuscoguttatus, larva. Bull. Pen. Perikanan, Spec. Edit. 1: 51–64.
Korniyenko, G. S., 1971. The role of infusoria in the food of larvae of phytophagous fishes. J. Ichthyol. 11: 241–246.
Lasker, R., 1978. The relation between oceanographic conditions and larval anchovy food in the California Current: identification of factors contributing to recruitment failure. Rapp. P.-v. Réun. Cons. int. Explor. Mer. 173: 212–230.
Lasker, R., H. M. Feder, G. H. Theilacker &; R. C. May, 1970. Feeding, growth, and survival of Engraulis mordax larvae reared in the laboratory. Mar. Biol. 5: 345–353.
Leakey, R. J. G., P. H. Burkill &; M. A. Sleigh, 1992. Planktonic ciliates in Southampton Water: abundance, biomass, production and role in pelagic carbon flow. Mar. Biol. 114: 67–83.
Lessard, E. J., M. P. Martin &; D. J. S. Montagnes, 1996. A new method for live-staining protists with DAPI and its application as a tracer of ingestion by walleye pollock (Theragra chalcogramma (Pallas)) larvae. J. exp. mar. Biol. Ecol. 204: 43–57.
Ohman, M. D., G. H. Theilacker &; S. E. Kaupp, 1991. Immunochemical detection of predation on ciliate protists by larvae of the northern anchovy (Engraulis mordax). Biol. Bull. 181: 500–504.
Pierce, R. W. &; J. T. Turner, 1992. Ecology of planktonic ciliates in marine food webs. Rev. Aquat. Sci. 6: 139–181.
Putt, M. &; D. K. Stoecker, 1989. An experimentally determined carbon: volume ratio for marine ‘oligotrichous’ ciliates from estuarine and coastal waters. Limnol. Oceanogr. 34: 1097–1103.
Sanders, R. W., 1987. Tintinnids and other microzooplankton-seasonal distributions and relationships to resources and hydrography in a Maine estuary. J. Plankton Res. 9: 65–77.
Sherr, E. B. &; B. F. Sherr, 1987. High rates of consumption of bacteria by pelagic ciliates. Nature 325: 710–711.
Sherr, E. B., B. F. Sherr &; G.-A. Paffenhöfer, 1986. Phagotrophic protozoa as food for metazoans: a ‘missing’ trophic link in marine pelagic food webs? Mar. Microb. Food Webs. 1: 61–80.
Stoecker, D. K. &; J. M. Capuzzo, 1990. Predation on protozoa: its importance to zooplankton. J. Plankton Res. 12: 891–908.
Stoecker, D. K., M. W. Silver, A. E. Michaels &; L. H. Davis, 1988. Obligate mixotrophy in Laboea strobila, a ciliate which retains chloroplasts. Mar. Biol. 99: 415–423.
Uye, S., N. Nagano &; H. Tamaki, 1996. Geographical and seasonal variations in abundance, biomass and estimated production rates of microzooplankton in the Inland Sea of Japan. J. Oceanogr. 52: 689–703.
Van der Meeren, T. &; T. Næss, 1993. How does cod (Gadus morhua) cope with variability in feeding conditions during early larval stages? Mar. Biol. 116: 637–647.
Verity, P. G. &; C. Langdon, 1984. Relationships between lorica volume, carbon, nitrogen and ATP content of tintinnids in Narragansett Bay. J. Plankton Res. 6: 859–868.
Watson, W. &; R. L. Davis Jr., 1989. Larval fish diets in shallow coastal waters off San Onofre, California. Fish. Bull. U.S.A. 87: 569–591.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagano, N., Iwatsuki, Y., Kamiyama, T. et al. Effects of marine ciliates on survivability of the first-feeding larval surgeonfish, Paracanthurus hepatus: laboratory rearing experiments. Hydrobiologia 432, 149–157 (2000). https://doi.org/10.1023/A:1004094825739
Issue Date:
DOI: https://doi.org/10.1023/A:1004094825739