Abstract
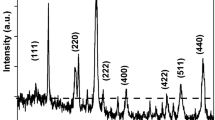
An electrochemical cell and relevant electrodes have been carried out in order to study in situ various electrochemical reactions involving iron using57Fe Mössbauer resonance. The electrochemical behaviour of iron in strongly alkaline solution (KOH 5N) in the potential range (−1200<E<−200 mV) has been examined. The introduction of sulfide ions in this electrolyte leads to the formation of a new phase, the mackinawite FeS. Optimized electrochemical conditions for growing a FeS deposit on an iron electrode have been specified.
Similar content being viewed by others
References
W.E. O'Grady and O'M. Bockris, Surf. Sci. 38 (1973) 249.
C. Fierro, R.E. Carbonio, D. Scherson and E.B. Yeager, J. Phys. Chem. 91 (1987) 6579.
U. Stumm, W. Meigel and P. Gütlich, Hyp. Int. 28 (1986) 923.
J. Elridge and R.W. Hoffman, J. Electrochem. Soc. 136 (1989) 955.
C. Chanson, L. Fournès an J-C. Grenier, C. R. Acad. Sci. Paris 303 (1986) 1633.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fournes, L., Grenier, JC., Chanson, C. et al. Use of in situ Mössbauer spectroscopy for electrochemical reactions involving57Fe. Hyperfine Interact 57, 1829–1832 (1990). https://doi.org/10.1007/BF02405729
Issue Date:
DOI: https://doi.org/10.1007/BF02405729