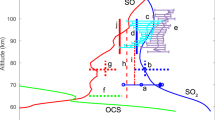
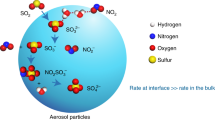
Abstract
The reaction between dimethyl sulphide (DMS) and the nitrate radical NO3 in dark air was studied in a Teflon bag, monitoring products formed in the gas phase together with aerosol composition and size distributions in the course of the experiment. The formation of the condensable products methane sulphonic acid (MSA) and sulphuric acid (H2SO4) was found to happen via a gaseous, relatively stable PAN-like peroxynitrate intermediate (CH3S(O)O2NO2 or CH3S(O)2O2NO2, called MSPN) which can build up to concentrations in the gas phase that are a multiple of MSA and H2SO4. A coupled gas chemistry-aerosol dynamics model was fitted to the experimental data and led to a consistent description of the partitioning of the S-containing products (SO2, MSPN, MSA and H2SO4) over gas and aerosol phase. The optimized chemical model reproduces adequately the observed strong NO dependence of the MSPN-to-(MSA+H2SO4) conversion rate by gas-phase reactions. The fitted loss rate for MSPN via pathways not included in the gas-phase mechanisms (e.g. reaction on aerosol particles or on the wall) is 100–500 times smaller than for N2O5. The model predicts further that about 50% of the initial DMS is transformed to SO2. Fitting the aerosol dynamics model to the observed aerosol growth rate, led to an estimate for the MSA condensation accommodation coefficient (αMSA>0.1) and for the MSA/H2SO4 formation ratio (1.2–3). The chemical model predicts that MSPN might be an important reservoir species for nitrogen, sulphur, and for aerosol formation in marine regions that are impacted by NO x -rich air masses.
Similar content being viewed by others
References
Andreae, M. O., Ferek, R. J., Bermond, F., Byrd, K. P., Engstrom, R. T., Hardin, S., Houmere, P. D., LeMarrec, F., Raemdonck, H., and Chatfield, R. B., 1985, Dimethyl sulfide in the marine atmosphere,J. Geophys. Res. 90, 12891–12900.
Andreae, M. O., 1990, The global biogeotechnical sulfur cycle: a review,Marine Chem. 30, 1–29.
Atkinson, R., Baulch, D. L., Cox, R. A., Hampson, R. F. Jr., Kerr, J. A., and Troe, J., 1989, Evaluated kinetic and photochemcial data for atmospheric chemistry: supplement III,J. Phys. Chem. Ref. Data, 881.
Atkinson, R. and Lloyd, A. C., 1984, Evaluation of kinetic and mechanistic data for modelling of photochemical smog,J. Phys. Chem. Ref. Data 13, 315.
Balla, R. J., Nelson, H. H., and McDonald, J. R., 1986, Kinetics of the reaction of CH3S with NO, NO2 and O2,Chem. Phys. 109, 101–107.
Barnes, I., Bastian, V., Becker, K. H., and Niki, H., 1987, FTIR spectroscopic studies of the CH3S + NO2 reaction under atmospheric conditions,Chem. Phys. Lett. 140, 451–457.
Barnes, I., Bonsang, B., Brauers, T., Carlier, P., Cox, R. A., Dorn, H. P., Jenkin, M. E., Le Bras, G., and Platt, U., 1991, Laboratory and field studies of oxidation processes occurring in the atmospheric marine boundary layer (Oceano-Nox CEC project), Commission of the European Communities, Air Pollution Research Report 35.
Bates, T. S., Cline, J. D., Gammon, R. H., and Kelly-Hansen, S. R., 1987, Regional and seasonal variations in the flux of oceanic dimethylsulphide to the atmosphere,J. Geophys. Res. 92, 2930–2938.
Covington, A. K., Robinson, R. A., and Thompson, R., 1973, Osmotic and activity coefficients for aqueous methane sulfonic acid solutions at 25C,J. Chem. Eng. Data 18, 422–423.
DeMore, W. B., Margitan, J. J., Molina, M. J., Watson, R. T., Golden, D. M., Hampson, R. F., Kurylo, M. J., Howard, C. J., and Ravishankara, A. R., 1985, Chemical kinetics and photochemical data for use in stratospheric modelling. No 7, JPL Publication.
DeMore, W. B., Sander, S. P., Golden, D. M., Molina, M. J., Hampson, R. F., Kurylo, M. J., Howard, C. J., and Ravishankara, A. R., 1990, Chemical kinetics and photochemical data for use in stratospheric modelling, No 9, JPL Publication 90–1.
DeMore, W. B., Sander, S. P., Golden, D. M., Hampson, R. F., Kurylo, M. J., Howard, C. J., Ravishankara, A. R., Kolb, C. E., and Molina, M. J., 1992, Chemical kinetics and photochemical data for use in stratospheric modelling, No 10, JPL Publication 92–20.
Dlugokencky, E. J. and Howard, C. J., 1988, Laboratory studies of NO3 radical reactions with some atmospheric sulphur compounds,J. Phys. Chem. 92, 1188–1193.
Fuchs, N. A., 1964,The Mechanics of Aerosols, Dover, New York, Chapter VII pp. 290–291.
Grosjean, D., 1985, Wall losses of gaseous pollutants in outdoor teflon chambers,Environ. Sci. Technol. 19, 1059–1065.
Hatakeyama, S., Izumi, K., and Akimoto, H., 1985, Yield of SO2 and formation of aerosol in the photo-oxidation of DMS under atmospheric conditions,Atmos. Environ. 19, 135–141.
Hoppel, W. A., 1987, Nucleation in the MSA-water vapour system,Atmos. Environ. 21, 2703–2709.
Jaecker-Viorol, A., Ponche, J. L., and Mirabel, P., 1990, Vapor pressure in the ternar system water-nitric acid-sulfuric acid at low temperatures,J. Geophys. Res. 95, 11857–11863.
Jensen, N. R., Hjorth, J., Lohse, C., Skov, H., and Restelli, G., 1991, Products and mechanism of the reaction between NO3 and dimethylsulphide in air,Atmos. Environ. 25, 1897–1904.
Jensen, N. R., Hjorth, J., Lohse, C., Skov, H., and Restelli, G., 1992, Products and mechanisms of the gas phase reactions of NO3 with CH3SCH3, CD3SCD3, CH3SH and CH3SSCH3,J. Atmos. Chem. 14, 95–108.
Jensen, N. R. and Hjorth, J., 1994, Evidence for the formation of a stable intermediate in the tropospheric oxidation of DMS,Geophys. Res. Lett., in press.
Kreidenweis, S. M., Flagan, R. C., Seinfeld, J. H., and Okuyama, K., 1989, Binary nucleation of methane sulfonic acid and water,J. Aerosol Sci. 20, 585–607.
Kusik, C. L. and Meissner, H. P., 1978, Electrolyte activity coefficients in inorganic processing,AIChe Symp. Ser. 173, 14–20.
Muzorkewich, M. and Calvert, J., 1988, Reaction probability of N2O5 on aqueous aerosols,J. Geophys. Res. 93, 15889–15896.
Nair, P. V. N. and Vohra, K. G., 1975, Growth of aqueous sulphuric acid droplets as a function of relative humidity,J. Aerosol Sci. 6, 265–271.
Renninger, R. G., Hiller, F. C., and Bone, R. C., 1981, Comment on ‘Self-nucleation in the sulfuric acid-water system’,J. Chem. Phys. 75, 1584.
Spiro, P. A., Jacob, D. J., and Logan, J. A., 1992, Global inventory of sulfur emissions with 1°×1° resolution,J. Geophys. Res. 97, 6023–6036.
Tyndall, G. S., Burrows, J. P., Schneider, W., and Moortgat, G. K., 1986, Rate coefficient for the reaction between NO3 radicals and dimethylsulphide,Chem. Phys. Lett. 130, 463–466.
Tyndall, G. S. and Ravishankara, A. R., 1989 Kinetics and mechanisms of the reactions of CH3S with O2 and NO2 at 298 K,J. Phys. Chem. 93, 2426–2435.
Tyndall, G. S. and Ravishankara, A. R., 1991, Atmospheric oxidation of reduced sulfur species,J. Phys. Chem. 23, 483–527.
Van Dingenen, R., Raes, F., and Vanmarcke, H., 1989, Molecule and aerosol particle wall losses in smog chambers made of glass,J. Aerosol Sci. 20, 113–122.
Van Dingenen, R. and Raes, F., 1991, Determination of the condensation accommodation coefficient of sulfuric acid on water-sulfuric acid aerosol,Aerosol Sci. Technol. 15, 93–106.
Van Dingenen, R. and Raes, F., 1993, Ternary nucleation of methane sulphonic acid, sulphuric acid and water vapour,J. Aerosol Sci. 24, 1–17.
Van Doren, J. M., Watson, L. R., Worsnop, D. R., Zahniser, M. S., and Kolb, C. E., 1991, Uptake of N2O5 and HNO3 by aqueous sulfuric acid droplets,J. Phys. Chem. 95, 1684–1689.
Wallington, Y. J., Atkinson, R., Winer, A. M., and Pitts, J. N. Jr., 1986a, Absolute rate constants for the gas-phase reactions of the NO3 radical with CH3SCH3, NO2, CO and a series of alkanes at 298±2 K,J. Phys. Chem. 90, 4640–4643.
Wallington, T. J., Atkinson, R., Winer, A. M., and Pitts, J. N. Jr., 1986b, Absolute rate constants for the gas-phase reactions of the NO3 radical with CH3SH, CH3SCH3, CH3SSCH3, H2S, SO2, and CH3OCH3 over the temperature range 280–350 K,J. Phys. Chem. 90, 5393–5396.
Warren, D. R. and Seinfeld, J. H., 1985, Nucleation and growth in a continuously reinforced vapor,Aerosol Sci. Technol. 3, 135–153.
Wayne, R. P., Barnes, I., Biggs, P., Cox, R. A., Hampson, R. F. Jr., Kerr, J. A., and Troe, J., 1991, The nitrate radical: physics, chemistry and the atmosphere,Atmos. Environ. 25, 1–212.
Wyslouzil, B. E., Seinfeld, J. H., Flagan, R. C., and Okuyama, K., 1991a, Binary nucleation in acid-water systems. I. Methanesolfonic acid-water,J. Chem. Phys. 94, 6827–6841.
Wyslouzil, B. E., Seinfeld, J. H., Flagan, R. C., and Okuyama, K., 1991b, Binary nucleation in acid-water systems. II: Sulfuric acid-water and a comparison with methanesulfonic acid-water,J. Chem. Phys. 94, 6842–6850.
Yin, F., Grosjean, D., Flagan, R., and Seinfeld, J. H., 1990, Photooxidation of dimethyl sulfide. II: Mechanism Evaluation,J. Atmos. Chem. 11, 365–399.
Zeleznik, F. J., 1991, Thermodynamic properties of the aqueous sulfuric acid system to 350 K,J. Phys. Chem. Ref. Data 20, 1157–1200.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van Dingenen, R., Jensen, N.R., Hjorth, J. et al. Peroxynitrate formation during the night-time oxidation of dimethylsulfide: Its role as a reservoir species for aerosol formation. J Atmos Chem 18, 211–237 (1994). https://doi.org/10.1007/BF00696780
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00696780