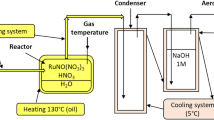
Abstract
Rutnenium volatilized from boiling nitric acid with an induction period, which depended on both the concentrations of nitric acid and ruthenium in the solution. The lower the nitric acid concentration, the longer the induction period. A high ruthenium concentration brought about a reduction of the induction period. The rate of volatilization of ruthenium did not directly depend on the concentration of nitric acid. The addition of 0.1M ferric ion to the ruthenium solution retarded volatilization rate of ruthenium to one twentieth under boiling of the solution.
Similar content being viewed by others
References
G. G. EICHHOLZ, Progr. Nucl. Energy, 2 (1978) 29.
A. S. WILSON, Rept. HW-45620, 1956.
A. S. WILSON, J. Chem. Eng. Data, 5 (1960) 521.
C. E. MAY, K. L. ROHDE, B. J. NEWBY, B. D. WITHERS, Rept. OD-14439, 1958.
J. M. FLETCHER, I. L. LENKINS, F. M. LEVER, F. S. MARTIN, A. R. POWELL, R. TODD, J. Inorg. Nucl. Chem., 1 (1955) 378.
T. SATO, J. Radioanal. Nucl. Chem., 104 (1986) 151.
Y. KODA, J. Radioanal. Chem., 6 (1970) 345.
T. TITANI, Inorganic Chemistry, Sangyoutosyo, Tokyo, 1964.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sato, T. Volatilization behaviour of ruthenium from boiling nitric acid. Journal of Radioanalytical and Nuclear Chemistry, Articles 129, 77–84 (1989). https://doi.org/10.1007/BF02037570
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02037570