Abstract
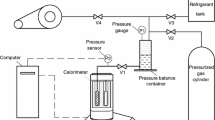
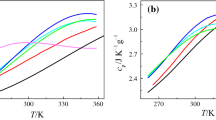
The constant pressure liquid-phase heat capacities of 21 hydrogen containing fluorinated propane and butane derivatives and one fluorinated ether (CF3OCF2H) with boiling points ranging from -34.6° to 76.7°C have been measured to 3% accuracy by differential scanning calorimetry at 40°C. The measurements are needed to help identify alternative refrigerants and blowing agents that do not deplete the stratospheric ozone layer. The DSC method has two significant advantages for this purpose, which are:
-
(i)
only small samples (less than 100 mg) are required, and
-
(ii)
the instruments are available in many laboratories and can be used for the heat capacity measurement of liquids with subambient boiling points without modification or special accessories.
Zusammenfassung
Mittels DSC bei 40°C wurden die Flüssigphasen-Wärmekapazitäten für konstanten Druck von 21 wasserstoffhaltigen fluorierten Propan- und Butanderivaten und von einem fluorierten Ether (CF3OCF2H) mit Siedenpunkten zwischen -34.6° und 76.7°C gemessen. Diese Messungen dienen der Suche nach alternativen Kühl- und Treibmitteln, welche die Ozonschicht der Stratosphäre nicht mindern. Für diese Aufgabe hat die DSC-Methode zwei eindeutige Vorteile:
-
(i)
es werden nur geringe Probenmengen benötigt (weniger als 100 mg) und
-
(ii)
die Geräte sind in vielen Laboratorien zugänglich und können ohne Ånderungen zur Messung von Wärmekapazitäten von Flüssigkeiten mit Siedepunkten unter Umgebungstemperatur verwendet werden.
Similar content being viewed by others
References
United Nations Environmental Programme (UNEP) 1987. Montreal Protocol on Substances that Deplete the Ozone Layer, Final Act, New York, United Nations.
Clean Air Act, Title VI — Stratospheric Ozone Protection, Public Law 101–549, November 15, 1990.
The number code used to designate compounds was devised by the American Society of Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE) for methane, ethane, and cyclo-alkane refrigerants [See Refrigerating Engineering, 65 (1957) 49]. The code was unofficially extended to include a wide variety of fluoroaliphatics by manufacturers of these chemicals.
United Nations Environment Programme (UNEP) Meeting, London, 1990.
A. L. Beyerlein, D. D. DesMarteau and S. H. Hwang, International CFC and Halon Alternatives Conference, Sponsored by the Alliance for Responsible CFC Policy, Baltimore, MD, December 3–5, 1991.
M. O. McLinden, J. S. Gallager, L. A. Weber, G. Morrison, D. Ward, A. R. H. Goodwin, M. R. Moldover, J. W. Schmidt, H. B. Chae, T. J. Bruno, J. F. Ely and M. L. Huber, ASHRAE Trans., 95 (1989) 263.
B. Wunderlich, Thermal Analysis, Academic Press, New York 1990.
W. Hemminger and G. Hohne, Calorimetry, Fundamentals and Practice, Verlag Chemie, Weinheim, Federal Republic of Germany 1984.
E. A. Turi, ed., “Thermal Characterization of Polymeric Materials”, Academic Press, New York 1982.
S. C. Mraw and D. F. Naas-O'Rourke, J. Chem. Thermodynam., 12 (1980) 691.
S. C. Mraw, J. L. Heidman, S. C. Hwang and C. Tsonopoulos, Ind. Eng. Chem. Process Des. Dev., 23 (1984) 577.
S. C. Mraw and D. F. Naas-O'Rourke, J. Chem. Thermodynamics, 13 (1981) 199.
M. Stephens and J. D. Olson, Thermochim. Acta, 76 (1984) 79.
R. C. Reid, I. M. Prausnitz and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York 1987.
Thermophysical Properties of Refrigerants, American Society of Heating, Refrigerating, and Air Conditioning Engineers, Inc., New York 1976.
D. W. Osborne, C. S. Garner, R. N. Doescher and D. M. Yost, J. Am. Chem. Soc., 63 (1941) 3496.
L. Riedel, Z. Gesamte Kalte-Ind., 46 (1939) 105.
R. C. Downing, Transport Properties of ‘Freon’ Fluorocarbons and Other Fluorinated Compounds, Freon Technical Bulletin C-30A, E. I. DuPont de Nemours and Co., Inc., 1966.
H. Suzuki and B. Wunderlich, J. Thermal Anal., 29 (1984) 1369.
U. Gaur, A. Mehta and B. Wunderlich, J. Thermal Anal., 13 (1978) 71.
B. I. Lee and M. G. Kesler, AIChE J., 21 (1975) 510.
R. E. Thek and L. I. Stiel, AIChE J., 12 (1966) 599.
R. E. Thek and L. I. Stiel, AlChe J., 13 (1967) 626.
R. W. Hankinson and G. H. Thomson, AIChE J., 25 (1979) 653.
L. Riedel, Chem. Ing. Tech., 26 (1954) 679.
B. Wunderlich, J. Thermal Anal., 32 (1987) 1949.
Y. Jin and B. Wunderlich, J. Thermal Anal., 36 (1990) 765, 1519.
Author information
Authors and Affiliations
Additional information
This work was supported jointly by the U.S. Environmental Protection Agency's Stratospheric Ozone Protection Branch at Research Triangle Park, North Carolina and the Electric Power Research Institute at Palo Alto, California.
The authors are grateful to the U. S. Environmental Protection Agency's Stratospheric Ozone Protection Branch, Research Triangle Park, NC and the Electric Power Research Institute, Palo Alto, CA, for supporting this research.
Rights and permissions
About this article
Cite this article
Hwang, S.H., DesMarteau, D.D., Beyerlein, A.L. et al. The heat capacity of fluorinated propane and butane derivatives by differential scanning calorimetry. Journal of Thermal Analysis 38, 2515–2528 (1992). https://doi.org/10.1007/BF01974629
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01974629