Abstract
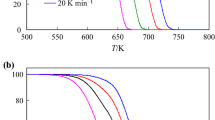
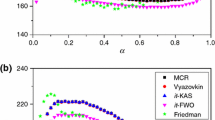
The thermal decomposition of Eu2(BA)6(bipy)2 (BA=C2H5N– 2, benzoate; bipy=C10H8N2, 2,2'-bipyridine)and its kinetics were studied under the non-isothermal condition by TG-DTG, IR and SEM methods. The kinetic parameters were obtained from analysis of the TG-DTG curves by the Achar method, the Madhusudanan-Krishnan-Ninan (MKN) method, the Ozawa method and the Kissinger method. The most probable mechanism function was suggested by comparing the kinetic parameters. The kinetic equation for the first stage can be expressed as: dα/dt=Aexp(–E/RT)3(1–α)2/3.
Similar content being viewed by others
REFERENCES
Y. Zhang, L. P. Jin and S. Z. Lü, J. Inorg. Chem. (in Chinese), 13 (1997) 280.
B. N. Achar, Proc. Int. Clay. Conf., Jerusalem, 1 (1966) 67.
P. M. Madhusudhanan, K. Krishman and K. N. Ninan, Thermochim. Acta, 97 (1986) 189.
T. Ozawa, Bull. Chem. Soc. Japan, 38 (1965) 1881.
H. E. Kissinger, Anal. Chem., 29 (1957) 17024.
J. Straszko, M. Olstak-Humienik and J. Mozejko, Thermochim. Acta, 292 (1997) 145.
Y. Z. Li, Thermal Analysis, Tsinghua University Press, Beijing, 1978, p. 94.
X. Gao and D. Dollimore, Thermochim. Acta, 215 (1993) 47.
R. Z. Hu, Z. Q. Yang and Y. J. Liang, Thermochim. Acta, 123 (1988) 135.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, JJ., Wang, RF., Li, JB. et al. Studies on Non-isothermal Kinetics of the Thermal Decomposition of Eu2(BA)6(bipy)2. Journal of Thermal Analysis and Calorimetry 62, 747–755 (2000). https://doi.org/10.1023/A:1026785727479
Issue Date:
DOI: https://doi.org/10.1023/A:1026785727479