Summary
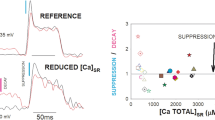
In adult skeletal muscle, G-proteins have been shown to modulate the calcium channels both directly and through a cAMP-dependent phosphorylating mechanism. We have investigated the action of G-proteins on the L-type calcium current in cultured rat muscle cells (myoballs) under voltage clamp in whole cell or perforated patch modes. Intracellular photolytic release of 200 μM GTPγS inhibited the L-type calcium current. Inclusion of 500 μM uncaged GTPγS in the patch pipette in the whole cell configuration reduced the calcium current by a similar amount. Under perforated patch conditions external application of 10 μM of the β-adrenergic agonist isoproterenol also reduced the calcium current. Pretreatment of the cells with pertussis toxin reversed the effect of GTPγS and removed that of isoproterenol. We conclude that rat myoballs contain β-adrenergic receptors that inhibit the L-type calcium current, and that this inhibition is mediated by a pertussis toxinsensitive G-protein.
Similar content being viewed by others
References
ARREOLA, J., CALVO, J., GARCIA, M. C. & SANCHEZ, J. A. (1987) Modulation of calcium channels of twitch skeletal muscle fibres of the frog by adrenaline and cyclic adenosine monophosphate. J. Physiol. (Lond.) 393, 307–30.
Bowman, W. C. & Nott, M. W. (1969) Action of sympathomimetic amines and their antagonists on skeletal muscle. Pharmac. Rev. 21, 27–72.
CARNEY, L. & DONALDSON, S. K. (1991) Inhibitory and excitatory GTPγS effects in mechanically skinned rabbit skeletal muscle fibres. Biophys. J. 59, 66A.
Cognard, C., Lazdunski, M. & Romey, G. (1986) Different types of Ca channels in mammalian skeletal muscle cells in culture. Proc. Natl. Acad. Sci. USA 83, 517–21.
Cognard, C., Rivet, M. & Raymond, M. (1990) The blockade of excitation-contraction coupling by nifedipine in patchclamped rat skeletal muscle cells in culture. Pflügers Arch. 416, 98–105.
Curtis, B. M. & Catterall, W. A. (1985) Phosphorylation of the calcium antagonist receptor of the voltage-sensitive calcium channel by cAMP-dependent protein kinase. Proc. Natl. Acad. Sci. USA 82, 2528–32.
Divirgilio, F., Salviati, G., Pozzan, T. & Volpe, P. (1986) Is a guanine nucleotide-binding protein involved in excitation-contraction coupling in skeletal muscle? EMBO J 5, 259–62.
Dolphin, A. C., Wootton, J. F., Scott, R. H. & Trentham, D. R. (1988) Photoactivation of intracellular guanosine triphosphate analogues reduces the amplitude and slows the kinetics of voltage-activated calcium channel currents in sensor neurones. Pflügers Arch. 411, 628–38.
Garcia, J., Aldeco, R. G. & Stefani, E. (1990) Charge movement and calcium currents in skeletal muscle fibres are enhanced by GTPγS. Pflügers Arch. 417, 114–16.
Gonzalez-Serratos, H., Hill, L. & Valle-Aguilera, R. (1981) Effects of catecholamines and cyclic AMP on excitation-contraction coupling in isolated skeletal muscle fibres of the frog. J. Physiol. (Lond.) 315, 267–82.
Hamill, O. P., Marty, A., Neher, E., Sakmann, B. & Sigworth, F. J. (1981) Improved patch-clamp techniques for high-resolution current recordings from cells and cell-free membrane patches. Pflügers Arch. 391, 85–100.
Horn, R. & Marty, A. (1988) Muscarinic activation of ionic currents measured by a new whole cell recording method. J. Gen. Physiol. 92, 145–59.
Lamb, G. D. (1991) Calcium channels or voltage sensors? Nature 352, 113.
Lew, W. Y. W., Hryshko, L. V. & Bers, D. M. (1991) Dihydropyridine receptors are primarily functional L-type calcium channels in rabbit ventricular myocytes. Circ. Res. 69, 1139–45.
LIU O. Y., KARPINSKIE., BENISHIN C. G. & PANG P. K. T. (1992) Phenylephrine increases L-type calcium channel current in neonatal rat ventricular cells. FASEB J 6, A394.
Rapp, G. & Guth, K. (1988) A low cost high intensity flash device for photolysis experiments. Pflügfers Arch. 411, 200–3.
Rios, E. & Pizarro, G. (1991) Voltage sensor of excitation-contraction coupling in skeletal muscle. Phys. Rev. 71, 849–908.
Rivet, M., Cognard, C. & Raymond, G. (1989) The slow inward calcium current is responsible for part of the contraction of patch-clamped rat myoballs. Pflügers Arch. 413, 316–18.
Romey, G., Garcia, L., Dimitriadou, V., Pincon-Raymond, M., Rieger, F. & Lazdunski, M. (1989) Ontogenesis and localization of Ca channels in mammalian skeletal muscle in culture and role in excitation-concentration coupling. Proc. Natl. Acad. Sci. USA 86, 2933–7.
Scherer, N. M., Toro, M., Entman, M. L. & Birnbaumer, L. (1978) G-protein distribution in canine cardiac sarcoplasmic reticulum and sarcolemma: comparison to rabbit skeletal muscle membranes and to brain and erythrocyte G-proteins. Arch. Biochem. Biophys. 259, 431–40.
Schmid, A., Renaud, J. F. & Lazdunski, M. (1985) Short-term and long-term effects of β-adrenergic effectors and cAMP on nitrendipine-sensitive voltage-dependent Ca2+ channels of skeletal muscle. J. Biol. Chem. 260, 13041–6.
Schultz, G., Rosenthal, W. & Hescheler, J. (1990) Role of G-proteins in calcium channel modulation. Ann. Rev. Physiol. 52, 275–92.
Somasundaram, B., Tregear, R. T. & Trentham, D. R. (1991) GTPγS causes contraction of skinned frog skeletal muscle via the DHP-sensitive Ca channels of sealed T-tubules. Pflügers Arch. 418, 137–43.
Toutant, M., Barhanin, J., Bockaert, J. & Rouot, B. (1988) G-proteins in skeletal muscle. Evidence for a 40 kDa pertussis-toxin substrate in purified transverse tubules. Biochem. J. 254, 405–9.
Toutant, M., Gabrion, J., Vandaele, S., Peraldi-Roux, S., Barhanin, J., Bockaert, J. & Rouot, B. (1990) Cellular distribution and biochemical characterization of G-proteins in skeletal muscle: comparative location with voltage-dependent calcium channels. EMBO J 9, 363–9.
Villaz, M., Robert, M., Carrier, L., Beeler, T., Rout, B., Toutant, M. & Dupont, Y. (1989) G-protein dependent potentiation of calcium release from sarcoplasmic reticulum of skeletal muscle. Cell Signalling 1, 493–506.
Yatani, A., Imoto, Y., Codina, J., Hamilton, S. L., Brown, A. M. & Birnbaumer, L. (1988) The stimulatory G-protein of adenylylcyclase, Gs, also stimulates dihydropyridine-sensitive Ca channels. J. Biol. Chem. 263, 9887–95.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Somasundaram, B., Tregear, R.T. Isoproterenol and GTPγS inhibit L-type calcium channels of differentiating rat skeletal muscle cells. J Muscle Res Cell Motil 14, 341–346 (1993). https://doi.org/10.1007/BF00123099
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00123099