Summary
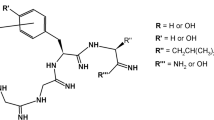
Dermorphin structure-activity relationships toward μ and δ opioid receptors were investigated using a series of synthetic peptides, in which the aromatic residues at positions 1 or/and 3 of the N-terminal tetrapeptide analogue H-Tyr-d-Arg-Phe-β-Ala-NH2 were replaced by unnatural or constrained amino acids.
Similar content being viewed by others
References
Broccardo, M., Erspamer, V., Erspamer, G.F., Improta, G., Linari, G., Melchiorri, P. and Montecucchi, P.C., Br. J. Pharmacol., 73 (1981) 625.
Montecucchi, P.C., de Castiglione, R., Piani, S., Gozzini, L. and Erspamer, V., Int. J. Pept. Protein Res., 17 (1981) 275.
Sasaki, Y., Ambo, A. and Susuki, K., Chem. Pharm. Bull., 39 (1991) 2316.
Tourwé, D., Verschueren, K., Frycia, A., Davis, P., Porreca, F., Hruby, V.J., Toth, G., Jaspers, H., Verheyden, P. and Van Binst, G., Biopolymers, 38 (1996) 1.
Castiglione-Morelli, M.A., Tancredi, T., Trivellone, E., Balboni, G., Marastoni, M., Salvadori, S., Tomatis, R. and Temussi, P., Biopolymers, 27 (1988) 1353.
Schiller, P.W., Weltrowska, G., Nguyen, T.M.-D., Lemieux, C., Chung, N.N., Marsden, B.J. and Wilkes, B.C., J. Med. Chem., 34 (1991) 3125.
Crescenzi, O., Amodeo, P., Cavicchioni, G., Guerrini, R., Picone, D., Salvadori, S., Tancredi, T. and Temussi, P., J. Pept. Sci., 2 (1996) 290.
Contarino, A., Zanotti, A., Drago, F., Natolino, F., Lipartiti, M. and Giusti, P., Arch. Pharmacol. (1998) in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Calderan, A., Ruzza, P., Ancona, B. et al. Synthesis, conformational and pharmacological studies on dermorphin N-terminal tetrapeptide analogues. Lett Pept Sci 5, 71–73 (1998). https://doi.org/10.1007/BF02443441
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02443441