Abstract
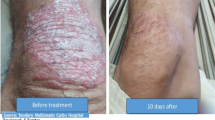
In anin vitro model, the release and penetration through a silicon membrane of different cream formulations and ointments containing 0.5% dithranol were studied. The results indicated that the method is not suitable for predictingin vivo release and penetration. Experiments with rabbits showed that the degree of skin irritation produced by a hospital-prepared product containing 0.5% dithranol was comparable to that of a commercial product. Clinical efficacy and side-effects of these two products were investigated in a double-blind left-right comparative study in 35 patients with chronic plaque psoriasis. The two creams proved to be equally effective and showed the same incidence of side-effects.
Similar content being viewed by others
References
Green PG, Kennedy CTC, Forbes DR. Anthralin stability in various vehicles. J Am Acad Dermatol 1987;16:984–8.
Shah VP, Elkins J, Lam S-Y, Skelly JP. Determination ofin vitro drug release from hydrocortisone creams. Int J Pharm 1989;53:53–9.
Martin B, Watts O, Shroot B, Jamoulle JC. A new diffusion cell — an automated method for measuring the pharmaceutical availability of topical dosage forms. Proceed Intern Symp Control Rel Bioact Mater 1988;15:374–5.
Seville RH. Dithranol-based therapies. In: Mier PD, Van der Kerkhof PCM, eds. Textbook of psoriasis. Edinburgh: Churchill Livingstone, 1986:178–89.
Fredriksson T, Petersson U. Severe psoriasis — oral therapy with a new retinoid. Dermatologica 1978;157: 238–44.
De Hoop D, Van Andel P, De Kort WJA. Psoriasis(dag)behandeling [Psoriasis (day) treatment]. Ned Tijdschr Geneeskd 1989;133:785–7.
De Hoop D, Van Andel P, De Kort WJA, Heinhuis J, Van Vloten WA, De Korte J. Behandeling van psoriasis in een dagbehandelingscentrum [Treatment of psoriasis in a day care centre]. Ned Tijdschr Geneeskd 1990;134:1220–3.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ros, J.J.W., Van der Meer, Y.G., De Hoop, D. et al. In vitro andin vivo comparison of creams containing dithranol 0.5%. Pharmaceutisch Weekblad Scientific Edition 13, 210–214 (1991). https://doi.org/10.1007/BF01988877
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01988877