Abstract
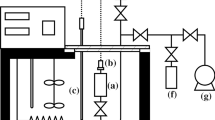
The critical temperature and pressure, vapor pressure, and PVT relations for gaseous and liquid 1-chloro-1,2,2,2-tetrafluoroethane (R124) were determined experimentally. The vapor pressure was measured in the temperature range from 278.15 K to the critical temperature. The PVT measurements were carried out using two types of volumeters in the temperature range from 278.15 to 423.15 K, at pressure up to 100 MPa. The numerical PVT data of gaseous state are fitted as a function of density to a modified Benedict-Webb-Rubin equation. The pressure-volume relations of the liquid at each temperature are correlated satisfactorily as a function of pressure by the Tait equation. The critical density and saturated vapor and liquid densities are also determined and some of the thermodynamic properties are derived from the experimental results.
Similar content being viewed by others
References
H. Kubota, S. Tsuda, M. Murata, T. Yamamoto, Y. Tanaka, and T. Makita, Rev. Phys. Chem. Jpn. 49:59 (1980).
D. Ambrose, J. Chem. Thermodynam. 10:765 (1978).
M. Uematsu, S. Saegusa, K. Watanabe, and I. Tanishita, Proc. 4th Int. Conf. High Press. (1974), p. 564.
A. Z. Wohl, Z. Phys. Chem. 99:234 (1921).
R. Bender, K. Bier, and G. Maurer, J. Chem. Thermodynam. 12:335 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kubota, H., Tanaka, Y., Makita, T. et al. Thermodynamic properties of 1-chloro-1,2,2,2-tetrafluoroethane (R124). Int J Thermophys 9, 85–101 (1988). https://doi.org/10.1007/BF00504002
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00504002