Abstract
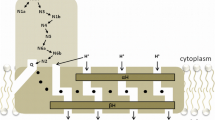
The components involved in the respiratory system of the euryarcheon Halobacterium salinarum were investigated by spectroscopic and polarographic techniques. Previous results about the cytochrome composition could be verified. However, under low oxygen tension, the expression of a d-type cytochrome was detected. Membranes exerted an NADH– and succinate–cytochrome-c oxidoreductase as well as an NADH and succinate oxidase activity. These activities could be blocked by the following inhibitors: 7-jodocarboxylic acid, giving evidence for the presence of a type II NADH dehydrogenase, antimycin A, and myxothiazol, indicating the presence of a complex III analog, and the typical succinate dehydrogenase (SDH) and terminal oxidase inhibitors. Complex I inhibitors like rotenone and annonine were inactive, clearly excluding the presence of a coupled NADH dehydrogenase. In addition, no [Fe-S] resonances in the region of the NADH dehydrogenase (NDH) clusters could be observed after NADH addition. One of the terminal oxidases could be shown to act as a cytochrome-c oxidase with a K m value of 37 μM and an activation energy of 23.7 kJ/mol. The relative molecular mass of the endogenous c-type cytochrome could be determined as 14.1 kD. The complex III analog could be enriched after detergent extraction with Triton X-100 and hydroxylapatite (HTP) chromatography. The partially purified complex contained a Rieske iron–sulfur cluster, b- and c-type cytochromes, and was catalytically active in the decylubiquinone–cytochrome-c oxidoreductase assay.
Similar content being viewed by others
REFERENCES
Aitken, D. M., and Brown, A. D. (1969). Biochim. Biophys. Acta 177, 351-354.
Anemüller, S., Lübben, M., and Schäfer, G. (1985). FEBS Lett. 193, 83-87.
Anemüller, S., Schmidt, C. L., Schäfer, G., and Teixeira, M. (1993). FEBS Lett. 318, 61-64.
Anemüller, S., Schmidt, C. L., Schäfer, G., Bill, E., Trautwein, A. X., and Teixeira, M. (1994). Biochem. Biophys. Res. Commun. 202, 252-257
Anemüller, S., Hettmann, T., Moll, R., Teixeira, M., and Schäfer, G. (1995). Eur. J. Biochem. 232, 563-568
Anemüller, S., Gomes, C., Teixeira, M., and Schäfer, G. (1996). In Proceedings of the 3rd European Conference on Bio-Inorganic Chemistry (Feiters, M. C., Hagen, W. R., and Veeger, C., eds.), NSR Center, Nijmegen, p. F7.
Belly, R. T., Bohlool, B. B., and Brock, T. D. (1973). Ann. N. Y. Acad. Sci. 225, 94-102.
Cheah, K. S. (1970a). Biochim. Biophys. Acta 216, 43-53.
Cheah, K. S. (1970b). Biochim. Biophys. Acta 197, 84-86.
Cheah, K. S. (1970c). Biochim. Biophys. Acta 205, 148-160.
Denda, K., Fujiwara, T., Seki, K., Yoshida, M., Fukumori, Y., and Yamanaka, T. (1991). Biochem. Biophys. Res. Commun. 181, 316-322.
Fujiwara, T., Fukumori, Y., and Yamanaka, T. (1987). Plant Cell Physiol. 28, 29-36.
Fujiwara, T., Fukumori, Y., and Yamanaka, T. (1989). J. Biochem. 105, 287-292.
Gabellini, N., and Hauska, G. (1983). FEBS Lett. 153, 146-150.
Gärtner, P. (1991). Eur. J. Biochem. 200, 215-222.
Gomez, C. M., and Teixeira, M. (1998). Biochem. Biophys. Res. Commun. 243, 412-415.
Hallberg, C., and Baltscheffsky, H. (1981). FEBS Lett. 125, 201-201.
Hallberg-Gradin, C., Hederstedt, L., and Baltscheffsky, H. (1985). Arch. Biochem. Biophys. 239, 200-205.
Hallberg-Gradin, and Colmsjö, A. (1989). Arch. Biochem. Biophys. 272, 130-136.
Hochstein, L. I., and Dalton, B. P. (1968). J. Bacteriol. 95, 37-42.
Holländer, R. (1978). J. Gen. Microbiol. 108, 165-167.
Iwasaki, T., Wakagi, T., and Oshima, T. (1995). J. Biol. Chem. 270, 30902-30908.
Janssen, S., Schäfer, G., Anemüller, S., and Moll, R. (1997). J. Bacteriol. 179, 5560-5569.
Kurowski, B., and Ludwig, B. (1987). J. Biol. Chem. 262, 13805-13811.
Laemmli, U. K. (1970). Nature 227, 680-695.
Lanyi, J. K. (1968). Arch. Biochem. Biophys. 128, 716-724.
Lanyi, J. K., and Stevenson, J. (1970). J. Biol. Chem. 245, 4074-4080.
Lübben, M. (1995). Biochim. Biophys. Acta 1229, 1-22.
Moll, R., and Schäfer, G. (1991). Eur. J. Biochem. 201, 593-600.
Oesterhelt, D., and Tittor, J. (1989). Trends Biochem. Sci. 14, 57-61.
Oettmeier, W., Masson, K., Soll, M., and Reil, E. (1994). Biochem. Soc. Trans. 22, 213-216.
Scharf, B., Wittenberg, R., and Engelhard, M. (1997). Biochemistry 36, 4471-4479.
Schäfer, G. (1996). Biochim. Biophys. Acta 1277, 163-200.
Schmidt, C. L., Anemüller, S, Teixeira, M., and Schäfer, G. (1995). FEBS Lett. 359, 239-243.
Schmidt, C. L., Anemüller, S., and Schäfer, G. (1996). FEBS Lett. 388, 43-46.
Smith, L., and Conrad, S. (1956). Arch. Biochem. Biophys. 63, 403-413.
Thomas, P. E., Ryan, D., and Levin, W. (1976). Anal. Biochem. 75, 168-176.
Trumpower, B. L., and Gennis, R. B. (1994). Annu. Rev. Biochem. 63, 675-716.
Wakao, H., Wakagi, T., and Oshima, T. (1987). J. Biochem. 102, 255-262.
Watters, C. (1978). Anal. Biochem. 88, 695-698.
Williams, J. N. (1964). Arch. Biochem. Biophys. 107, 537-543.
Rights and permissions
About this article
Cite this article
Sreeramulu, K., Schmidt, C.L., Schäfer, G. et al. Studies of the Electron Transport Chain of the Euryarcheon Halobacterium salinarum: Indications for a Type II NADH Dehydrogenase and a Complex III Analog. J Bioenerg Biomembr 30, 443–453 (1998). https://doi.org/10.1023/A:1020538129400
Issue Date:
DOI: https://doi.org/10.1023/A:1020538129400