Abstract
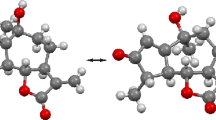
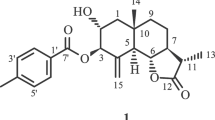
The sesquiterpene guayulin B, C23H30O3, 1, crystallizes with two independent molecules in monoclinic space group P21 with a = 18.099 (1), b = 6.0649(6), c = 19.126(1) Å, β = 100.82(1)°, V = 2062.1(5) Å3, and Z = 4. The double bonds of the cyclodecadiene ring are E, both methyl groups on the 10-membered ring are β-oriented. The cyclopropane ring at C6 and C7 also has the β orientation, while the anisoyloxy group is oriented α. The 10-membered rings of the two independent molecules have the same conformation as that of guayulin A.
Similar content being viewed by others
References
Watkins, S.F.; Fronczek, F.R.; Chiari, G.; Reynolds, G.W.; Rodriguez, E. J. Nat. Prod. 1985, 48, 631.
Schloman, W.W., Jr.; Hively, R.A.; Krishen, A.; Andrews, A.M. J. Agric. Food Chem. 1983, 31, 873.
Rodriguez, E.; Reynolds, G.W.; Thompson, J.A. Science 1981, 211, 1444.
Schloman, W.W., Jr.; Garrot, D.J., Jr.; Ray, D.T.; Bennett, D.J. J. Agric. Food Chem. 1986, 34, 177.
Sheldrick, G.M. SHELX86. Program for the Solution of Crystal Structures; University of Göttingen; Germany, 1985.
Fair, C.K. MolEN, An Interactive Structure Solution Procedure; Delft: The Netherlands, 1990.
Fischer, N.H.; Olivier, E.J.; Fischer, H.D. In Progress in the Chemistry of Organic Natural Products; Herz, W.; Grisebach, H.; Kirby, G.W., Eds.; Springer Verlag: New York, 1979; Vol. 38, p 47.
Castañeda-Acosta, J.; Pentes, H.G.; Fronczek, F.R.; Fischer, N.H. J. Chem. Crystallogr. 1997, 27, 635.
Crevoisier, M.; Steudle, K.C.; Buergi, H.B. Acta Crystallogr. 1984, C40, 979.
Martinez, M.; Flores, G.; Romo de Vivar, R.; Reynolds, G.; Rodriguez, E. J. Nat. Prod. 1986, 49, 1102.
Nishimura, K.; Hirobe, I.; Tori, K. Tetrahedron 1973, 29, 271.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Castañeda-Acosta, J., Fronczek, F.R. & Fischer, N.H. Guayulin B. Journal of Chemical Crystallography 29, 107–109 (1999). https://doi.org/10.1023/A:1009587718394
Issue Date:
DOI: https://doi.org/10.1023/A:1009587718394