Abstract
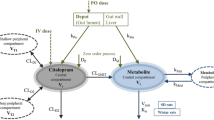
Sulfaethidole distribution and elimination in the rat was studied over a 90-fold dose range. This experimental design produced marked nonlinearity in the binding of Sulfaethidole to proteins in both interstitial fluid and plasma. Using a multicompartmental model consisting of binding of Sulfaethidole to plasma and interstitial fluid proteins, Sulfaethidole distribution in the body could be simulated. Urinary and biliary elimination of Sulfaethidole depended on the unbound drug mass in the plasma and urine flow. The results confirm the central role of the unbound species in the distribution and elimination of drugs with marked binding to plasma proteins.
Similar content being viewed by others
Abbreviations
- A 1 :
-
amount of drug in plasma (mg)
- A 2 :
-
amount of drug in interstitial fluid (mg)
- A 3 :
-
amount of drug in poorly perfused tissues (mg)
- A 4 :
-
amount of drug in highly perfused tissues (mg)
- fu 1 :
-
fraction of total drug in plasma unbound (dimensionless)
- fu 2 :
-
fraction of total drug in interstitial fluid unbound (dimensionless)
- fb 11 :
-
fraction of drug bound to first binding site on plasma protein (dimensionless)
- fb 12 :
-
fraction of drug bound to second binding site on plasma protein (dimensionless)
- fb 21 :
-
fraction of drug bound to first binding site on interstitial fluid protein (dimensionless)
- fb 22 :
-
fraction of drug bound to second binding site on interstitial fluid protein (dimensionless)
- K d,1 :
-
apparent dissociation constant of first binding site on protein (dimensionless)
- K d,2 :
-
apparent dissociation constant of second binding site on protein (dimensionless)
- B −1max,11 :
-
inverse of maximal binding capacity of first binding site on plasma protein (ml/mg)
- B 12−1max :
-
inverse of maximal binding capacity of second binding site on plasma protein (ml/mg)
- B −1max,21 :
-
inverse of maximal binding capacity of first binding site on interstitial fluid protein (ml/mg)
- B −1max,22 :
-
inverse of maximal binding capacity of second binding site on interstitial fluid protein (ml/mg)
- k 12 :
-
fractional transport rate of unbound drug from plasma to interstitial fluid (h−1)
- k 21 :
-
fractional transport rate of unbound drug from interstitial fluid to plasma (h−1)
- k 23 :
-
fractional transport rate of unbound drug from interstitial fluid to poorly perfused tissues (h−1)
- k 32 :
-
fractional transport rate of unbound drug from poorly perfused tissues to interstitial fluid (h−1)
- k 10 :
-
fractional rate of elimination of unbound drug from plasma (h−1)
- k 010 :
-
value during the first 210 min
- k 110 :
-
value after 270 min, linearly increased between 210 and 270 min (Eq. 5)
- fup t :
-
fraction of instantaneous binding of drug between plasma unbound drug and highly perfused tissue (dimensionless)
- V p :
-
plasma volume (ml)
- V is :
-
interstitial fluid volume (ml)
References
E. Krüger-Thiemer, W. Diller, L. Dettli, P. Bunger, and J. Seydel. Demonstration des Einflusses der Eiweissbindung und der Ionisation auf die Pharmacokinetik am kombinierten gaskinetischen Modell nach van't Hoff und Langmuir.Antibiot. Chemother. (Basel)12:171–193 (1964).
E. Krüger-Thiemer, W. Diller, and P. Bunger. Pharmacokinetic models regarding protein binding of drugs.Antimicrob. Agents Chemother. (1965/1966):183–191.
E. Krüger-Thiemer. Die Lösung pharmakologischer Probleme durch Rechenautomaten.Arzneim.-Forsch. 16:1431–1442 (1966).
J. G. Wagner. A new generalized nonlinear pharmacokinetic model and its implications. InBiopharmaceutics and Relevant Pharmacokinetics, 1st ed., Drug Intelligence Publications, Hamilton, Ill., 1971, pp. 252–259, 302–317.
J. G. Wagner. Nonlinear pharmacokinetics. Pharmacokinetics Seminar—Course II in Nonlinear Pharmacokinetics, June 19–21, 1972, at Northland Inn, Southfield, Mich.
J. G. Wagner. A modern view of pharmacokinetics.J. Pharmacokin. Biopharm. 1:363–401 (1973).
K. B. Bischoff, R. L. Dedrick, D. S. Zaharko, and J. A. Longstreth. Methotrexate pharmacokinetics.J. Pharm. Sci. 60:1128–1133 (1971).
R. L. Dedrick, D. S. Zaharko, and R. J. Lutz. Transport and binding of methotrexate in vivo.J. Pharm. Sci. 62:882–890 (1973).
A. R. DiSanto and J. G. Wagner. Pharmacokinetics of highly ionized drugs III: Methylen blue-blood levels in the dog and tissue levels in the rat following intravenous administration.J. Pharm. Sci. 61:1090–1094 (1972).
N. Gerber and J. G. Wagner. Explanation of dose-dependent decline of diphenylhydantoin plasma levels by fitting to the integrated form of the Michaelis-Menten equation.Res. Commun. Chem. Pathol. Pharmacol. 3:445–466 (1972). Cited in J. G. Wagner. A modern view of pharmacokinetics.J. Pharmacokin. Biopharm. 1:363–401 (1973).
R. J. K. Julkunen, M. Kekki, J.-J. Himberg, and B. Wahlström. Non-linear multicompartment model for drug binding by extracellular proteins with an application to warfarin.Acta Pharmacol. (KbH.)38:90–103 (1976).
M. Kekki, R. J. K. Julkunen, and B. Wahlström. Distribution pharmacokinetics of warfarin in the rat, a non-linear multicompartment model.Naunyn-Schmiedeberg's Arch. Pharmacol. 297:61–73 (1977).
A. Anton. The relation between the binding of sulfonamides to albumine and their antibacterial efficacy.J. Pharmacol. Exp. Ther. 129:282–290 (1960).
P. Pentikäinen, M. Kekki, and O. Mustala. Metabolism of14C-5HT in experimental liver cirrhosis in the rat.Scand. J. Gastroenterol. 4(suppl. 4):1–33 (1969).
A. C. Bratton and E. K. Marshall. A new coupling component for sulfanilamide determination.J. Biol. Chem. 128:537–550 (1939).
M. Kekki. Serum protein turnover in experimental hypo- and hyperthyroidism.Acta Endocrinol. 46(Suppl. 91) (1964).
H. Pohjanpalo. System identifiability based on the power series expansion of the solution.Math. Biosci. 41:21–33 (1978).
M. Berman. Kinetic analysis of turnover data. In S. Eisenberg (vol. ed.), R. Paoletti (ser. ed.),Intravascular Metabolism of Lipoproteins. Prog. Biochem. Pharmacol, Vol. 15, S. Karger, Basel, 1979, pp. 67–108.
H. Pohjanpalo. Description of the interactive real-time simulation program IRS. Technical Research Centre of Finland, Electrical Engineering Laboratory, publication series no. 34. VTT Offsetpaino 624/12, Espoo, Finland, 1978.
E. L. Foltz, J. V. Swintosky, and M. J. Robinson. Absorption, distribution and fate of sulfaethylthiadiazole administered orally and intravenously.Fed. Proc. 15:422 (1956).
J. V. Swintosky, M. J. Robinson, E. I. Folz, and S. M. Free. Sulfaethylthiadiazole I. Interpretations of human blood level concentrations following oral doses.J. Am. Pharm. Assoc. 46:399–403 (1957).
M. K. Husain, W. M. Manger, R. Weiss, C. Hart, and A. G. Franz. Vasopressin release in the rat after manual restraint: an effect of abdominal compression and not of “stress.” Proc. V Int. Congr. Endocrinol., 1976, p. 197. Cited in M. L. Forsling,Anti-Diuretic Hormone. Annual Research Reviews, Vol. 2, Churchill Livingstone, Edinburgh, 1977, pp. 78–79.
R. J. Lutz, R. L. Dedrick, J. A. Straw, M. M. Hart, P. Klubes, and D. S. Zaharko. The kinetics of methotrexate distribution in spontaneous canine lymphosarcoma.J. Pharmacokin. Biopharm. 3:77–97 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kekki, M., Julkunen, R.J.K. & Pohjanpalo, H. Pharmacokinetics of sulfaethidole in the rat: Nonlinear multicompartment solution. Journal of Pharmacokinetics and Biopharmaceutics 10, 27–51 (1982). https://doi.org/10.1007/BF01059182
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059182