Abstract
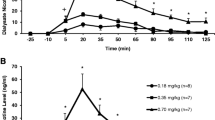
The tissue partition of cotinine was measured by a GC-MS method following a 6-day constant-rate input of nicotine and cotinine to male rats by means of an osmotic minipump. The tissue-to-blood partition coefficients of cotinine were calculated for adipose (0.08), brain (0.48), heart muscle (0.55), intestinal (0.53), hepatic (0.64), pulmonary (0.50), renal (0.99), and skeletal muscle tissue (0.51), following the cotinine infusion. When nicotine was infused the tissue partitioning of cotinine increased by a factor of 2.3–4.9, depending on the tissue sampled. Another group of animals were killed at timed intervals from 10 min to 30 hr, after having received a single intravenous bolus dose of 0.5 mg cotinine, and the washout of cotinine was traced in blood and tissues. A physiological model was used to simulate the disposition of cotinine. Generally, the model-predicted concentrations were consistent with those found experimentally. The fractional uptake of cotinine into various tissues was simulated. Blood, intestinal, and skeletal muscle tissues embodied more than 70% of the total body load of the drug. Clearance (Cl),volume of distribution (Vd),and the biological half-life (t1/2)were calculated both from the infusion study and by fitting a monoexponential model to the iv blood data of the rat. Significant differences were found in the apparent clearance calculated from the single iv bolus dose compared to the constant rate infusion. The volume of distribution was, however, consistent from both studies. The impact of a change in clearance was also simulated.
Similar content being viewed by others
References
A. J. Cohen and F. J. C. Roe.Monograph on the Pharmacology and Toxicology of Nicotine, Tobacco Advisory Council, Occasional Paper, London, 1981.
U.S. Public Health Service.The Health Consequences of Smoking: A Report of the Surgeon General (DHEW Publication No. HSM 72-7516), U.S. Government Printing Office, Washington, DC, 1972.
U.S. Public Health Service.The Health Consequences of Smoking: A Report of the Surgeon General (DHEW Publication No. HSM 71-7513, U.S. Government Printing Office, Washington, DC, 1971.
N. L. Benowitz. Clinical pharmacology of nicotine.Ann. Rev. Med. 37:21–32 (1986).
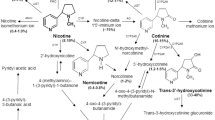
J. W. Gorrod and P. Jenner. The metabolism of tobacco alkaloids.Essays Toxicol. 6:35 (1975).
D. M. Turner, A. K. Armitage, R. H. Briant, and C. T. Dollery. Metabolism of nicotine by the isolated perfused dog lung.Xenobiotica 5:539 (1975).
N. L. Benowitz, F. Kuyt, P. Jacobs, III, R. T. Jones, and A-L. Osman. Cotinine disposition and effects.Clin. Pharmacol. Ther. 34:604–611 (1983).
L. Jansky and J. S. Hart. Cardiac output and organ blood flow in warm- and cold-acclimated rats exposed to cold.Can. J. Physiol. Pharmacol. 46:653–659 (1968).
K. B. Bischoff. Some fundamental considerations of the applications of pharmacokinetics to cancer chemotherapy.Cancer Chemother. Rep. (Part I)59:777–794 (1975).
J. Gabrielsson. Modeling of drugs in pregnancy: The application of pharmacokinetic principles and physiological flow models. Ph.D. thesis, University of Uppsala, 1985.
L. S. Goodman and A. Gilman.The Pharmacological Basis of Therapeutics, 6th ed., Macmillan, New York, 1980, p. 214.
P. Dominiak, G. Fuchs, S. von Toth, and H. Grobecker. Effects of nicotine and its metabolites on blood pressure in anaesthetized rats.Klin. Wochenschr. 63:90–92 (1985).
D. S. Riggs.The Mathematical Approach to Physiological Problems, MIT Press. Cambridge, MA, 1963.
K. S. Pang and M. Rowland. Hepatic clearance of drugs. I. Theoretical considerations of a “well-stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma and blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance.J. Pharmacokin. Biopharm. 5:625–653 (1977).
ACSL: Advanced Continuous Simulation Language. Reference Manual, 4th ed., Mitchell and Gauthier, Concord, MA, 1986.
M. Gibaldi and D. Perrier.Pharmacokinetics, 2nd ed., Marcel Dekker, New York, 1982.
M. Hakman and J. Gabrielsson.A User's Guide to MAXFIT, The Biomedical Center, Uppsala University, 1986.
N. L. Benowitz, R. P. Forsyth, K. L. Melmon, and M. Rowland. Lidocaine disposition kinetics in monkey and man I: Prediction by a perfusion model.Clin. Pharmacol. Ther. 16:87–98 (1974).
K. S. Rotenberg and J. Adir. Pharmacokinetics of nicotine in rats after muitiple-cigarette smoke exposure.Toxicol. Appl. Pharmacol. 69:1–11 (1983).
G. Lam, M. L. Chen, and W. L. Chiou. Determination of tissue to blood partition coefficients in physiologically based pharmacokinetic studies.J. Pharm. Sei. 71:454–456 (1982).
U.S. Public Health Service.Smoking and Health. A Report to the Surgeon General (DHEW Publication No. PHS 79-50066), U.S. Government Printing Office, Washington, DC, 1979.
G. A. Kyerematen, G. T. Passananti, F. E. Greene, and E. S. Vesell. Time-dependent induction of hepatic drug metabolism in rats by cotinine.Life Sci. 32:551–556 (1983).
W. H. Pitlick and R. H. Levy. Time-dependent kinetics: I. Exponential auto-induction of carbamazepine in monkeys.J. Pharm. Sci. 66:647–649 (1977).
C. M. Berlin and R. T. Schimke. Influences of turnover rates on the responses of enzymes to cortisone.Mol Pharmacol. 1:149 (1965).
P. J. De Schepper, A. Van Hecken, P. Daenens, and J. M. van Rossum. Kinetics of cotinine after oral and intravenous administration to man.Eur. J. Clin. Pharmacol. 31:583–588 (1978).
Author information
Authors and Affiliations
Additional information
The financial support from the Brown and Williamson Tobacco Company (USA) and the Åke Wiberg Foundation (Sweden) is gratefully acknowledged.
Rights and permissions
About this article
Cite this article
Gabrielsson, J., Boniesson, U. Constant-rate infusion of nicotine and cotinine. I. A physiological pharmacokinetic analysis of the cotinine disposition, and effects on clearance and distribution in the rat. Journal of Pharmacokinetics and Biopharmaceutics 15, 583–599 (1987). https://doi.org/10.1007/BF01068414
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01068414